2
answers
0
watching
58
views
11 Dec 2019
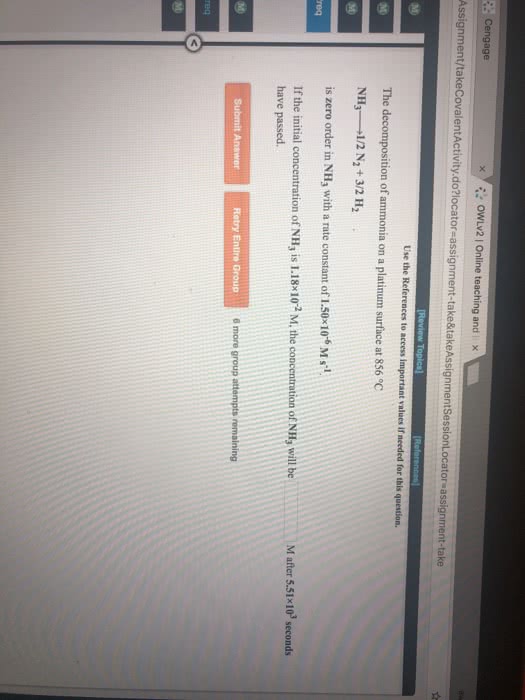
The decomposition of ammonia is found to be a first-order process with a rate constant k=0.0033s-1
2NH3 (g) ---> N2 (g) + 3H2 (g)
A) If the concentration of ammonia is initially 0.500 moles/L, then how long will it take for the concentration to decrease to 0.100 moles/L
B) What is the initial concentration of ammonia if the concentration is measured and found to be 0.25 moles/L after 30 seconds
C) What is the half life for this reaction?
The decomposition of ammonia is found to be a first-order process with a rate constant k=0.0033s-1
2NH3 (g) ---> N2 (g) + 3H2 (g)
A) If the concentration of ammonia is initially 0.500 moles/L, then how long will it take for the concentration to decrease to 0.100 moles/L
B) What is the initial concentration of ammonia if the concentration is measured and found to be 0.25 moles/L after 30 seconds
C) What is the half life for this reaction?
Read by 1 person
jagdishkumarLv7
17 Jul 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Reid WolffLv2
13 Dec 2019
Get unlimited access
Already have an account? Log in