2
answers
0
watching
13
views
13 Dec 2019
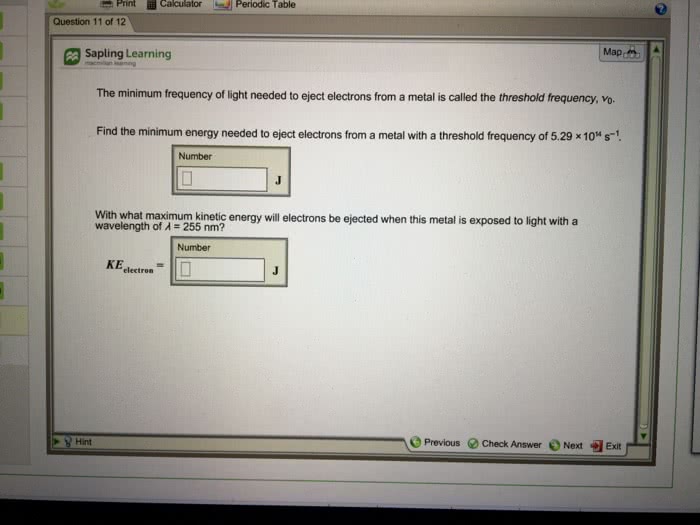
When light of frequency 4.96Ã1015 s-1 shines on the surface of a metal, electrons are ejected with a maximum kinetic energy of 3.88Ã10-19 J.
a) Calculate the wavelength of this light.
b) Find the binding energy of electrons to the metal.
c) What is the longest wavelength of light that will eject electrons from this metal?
When light of frequency 4.96Ã1015 s-1 shines on the surface of a metal, electrons are ejected with a maximum kinetic energy of 3.88Ã10-19 J.
a) Calculate the wavelength of this light.
b) Find the binding energy of electrons to the metal.
c) What is the longest wavelength of light that will eject electrons from this metal?
1 Oct 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Keith LeannonLv2
17 Dec 2019
Get unlimited access
Already have an account? Log in