2
answers
0
watching
5
views
13 Dec 2019
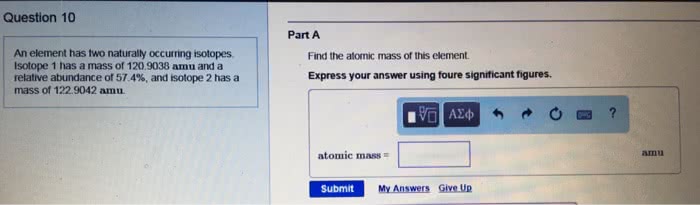
The element copper has two naturally occurring isotopees. One of these has a mass of 62.93 amu and a natural abundance of 69.09%. A. Second isotope has a mass of 64.9278 amu and a natural abundance of 30.91% . Calculate the atomic mass of copper.
The element copper has two naturally occurring isotopees. One of these has a mass of 62.93 amu and a natural abundance of 69.09%. A. Second isotope has a mass of 64.9278 amu and a natural abundance of 30.91% . Calculate the atomic mass of copper.
28 Jun
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Sixta KovacekLv2
17 Dec 2019
Get unlimited access
Already have an account? Log in