2
answers
0
watching
54
views
13 Dec 2019
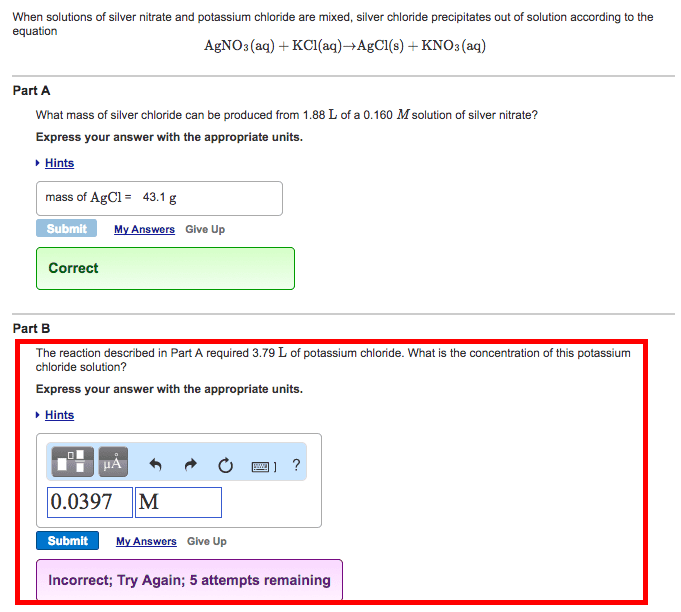
Aqueous solutions of potassium chloride and silver nitrate are mixed together. Which statement is correct?
A. 2 precipitates form
B. A gas is released
C. AgCl will precipitate from solution
D. KNO3 will precipitate from solution
E. No reaction will occur
Aqueous solutions of potassium chloride and silver nitrate are mixed together. Which statement is correct?
A. 2 precipitates form
B. A gas is released
C. AgCl will precipitate from solution
D. KNO3 will precipitate from solution
E. No reaction will occur
huyenanhhLv1
13 Jul 2022
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
Nelly StrackeLv2
17 Dec 2019
Get unlimited access
Already have an account? Log in