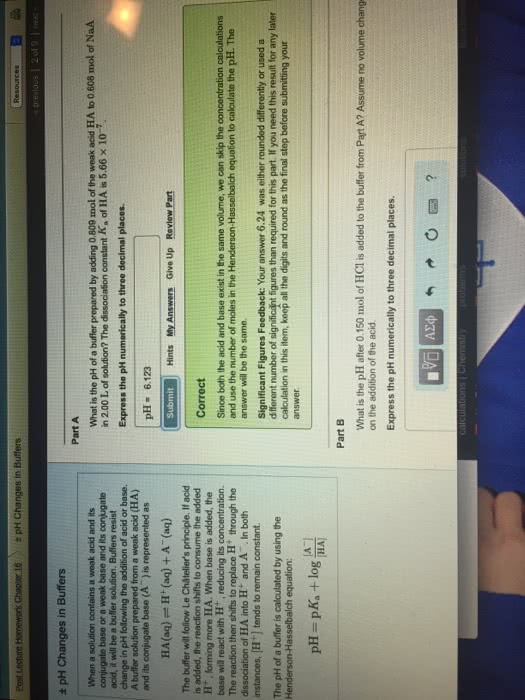
Problem 6.34
Titanium metal requires a photon with a minimum energy of6.94Ã10â19J to emit electrons.
Part A
What is the minimum frequency of light necessary to emit electrons from titanium via the photoelectric effect?
Express your answer using three significant figures.
ν = 1.05Ã1015 sâ1
SubmitMy AnswersGive Up
Correct
Significant Figures Feedback: Your answer 1.04â 1015 = 1.04Ã1015 sâ1 was either rounded differently or used a different number of significant figures than required for this part.
Part B
What is the wavelength of this light?
Express your answer using three significant figures.
λ = 286 nm
SubmitMy AnswersGive Up
Correct
Significant Figures Feedback: Your answer 288 nm was either rounded differently or used a different number of significant figures than required for this part.
Part C
Is it possible to eject electrons from titanium metal using visible light?
Is it possible to eject electrons from titanium metal using visible light? Yes, it is possible. No, it is impossible.
SubmitMy AnswersGive Up
Correct
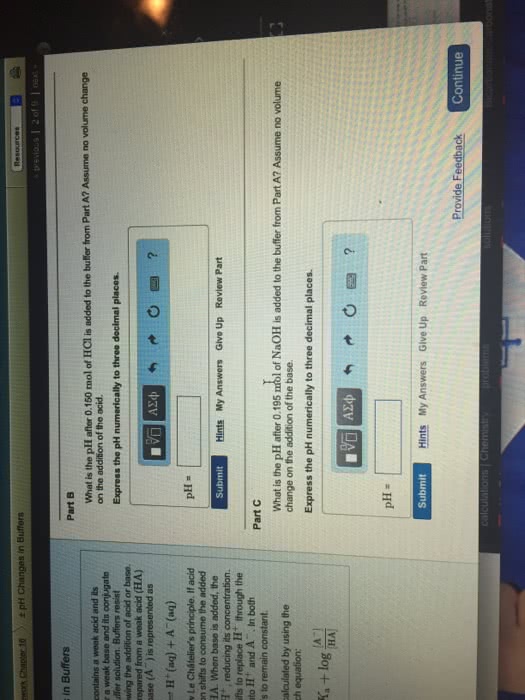
Part D
If titanium is irradiated with light of 233 nm, what is the maximum possible kinetic energy of the emitted electrons?
Express your answer using three significant figures.
E = 1.589â 10â19 J
SubmitMy AnswersGive Up
Correct
Part E
What is the maximum number of electrons that can be freed by a a burst of light whose total energy is 2.00 μJ.
Express your answer using three significant figures.
N = electrons
| Problem 6.34 Titanium metal requires a photon with a minimum energy of6.94Ã10â19J to emit electrons. | Part A What is the minimum frequency of light necessary to emit electrons from titanium via the photoelectric effect? Express your answer using three significant figures.
SubmitMy AnswersGive Up Correct Significant Figures Feedback: Your answer 1.04â 1015 = 1.04Ã1015 sâ1 was either rounded differently or used a different number of significant figures than required for this part. Part B What is the wavelength of this light? Express your answer using three significant figures.
SubmitMy AnswersGive Up Correct Significant Figures Feedback: Your answer 288 nm was either rounded differently or used a different number of significant figures than required for this part. Part C Is it possible to eject electrons from titanium metal using visible light? Is it possible to eject electrons from titanium metal using visible light?
SubmitMy AnswersGive Up Correct Part D If titanium is irradiated with light of 233 nm, what is the maximum possible kinetic energy of the emitted electrons? Express your answer using three significant figures.
SubmitMy AnswersGive Up Correct Part E What is the maximum number of electrons that can be freed by a a burst of light whose total energy is 2.00 μJ. Express your answer using three significant figures.
|