Alkane halogenation is a two-step reaction, as shown below. Using the table of bond dissociation energies, calculate the enthalpy of each step and the enthalpy of the overall reaction.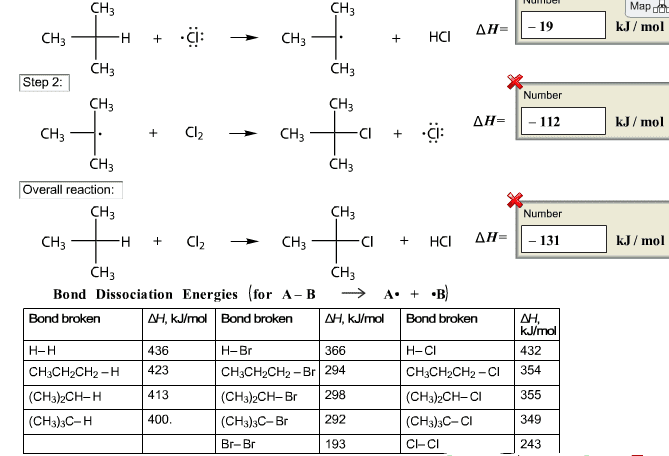
Map dct k.J/ mol CH3 CH3 ÎÎ-11-19 HCI CH3 CH3 CH3 Step 2 Number CH3 CH3 ÎÎ-11-112 k.J/ mol Cl + -Cl: CH3 CH3 CH3 Overall reaction CH3 CH3 Number CI HCI AH1 CH3 - 131 k.J / mol CH CH3 Bond Dissociation Energies for A-B A .B NH, kJ/moBond broke AH, kJlmol Bond broken AH, k/mol Bond broken 432 H-Br CH3CH2CH2-Br 29.4 CH3)2CH-Br 298 (CH3)3C-Br Br-Br H-Cl CH3CH2CH2-CI 354 (CH3)2CH-CI (CH3)3C-Cl C-CI 366 436 CH3CH2CH2- 423 413 400 H-H (CH3)2CH-H (CHs),C-H 355 349 243 292 193
Show transcribed image text Map dct k.J/ mol CH3 CH3 ÎÎ-11-19 HCI CH3 CH3 CH3 Step 2 Number CH3 CH3 ÎÎ-11-112 k.J/ mol Cl + -Cl: CH3 CH3 CH3 Overall reaction CH3 CH3 Number CI HCI AH1 CH3 - 131 k.J / mol CH CH3 Bond Dissociation Energies for A-B A .B NH, kJ/moBond broke AH, kJlmol Bond broken AH, k/mol Bond broken 432 H-Br CH3CH2CH2-Br 29.4 CH3)2CH-Br 298 (CH3)3C-Br Br-Br H-Cl CH3CH2CH2-CI 354 (CH3)2CH-CI (CH3)3C-Cl C-CI 366 436 CH3CH2CH2- 423 413 400 H-H (CH3)2CH-H (CHs),C-H 355 349 243 292 193 


