CHEM 1000 Lecture : D30137.docx
30 views1 pages
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related Documents
Related Questions
The formaldehyde molecule is shown below, and we will assumethat it is in the xy plane.
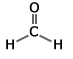
Which orbitals on carbon are involved in the sp2 hybrid orbitalsand which orbital is involved in ??
| 2s, 2pzinvolved in hybridization and2pxinvolved in ? bonding. | |
| The 2s, 2px, 2pyare involvedin hybridization and 2pzis involved in ? bonding. | |
| 2s, 2pxinvolved in hybridization and2pyinvolved in ? bonding. | |
| 2s, 2px, 2pzinvolved inhybridization and 2pyinvolved in ? bonding. |

