CHM136H1 Lecture : Tutorial 5.pdf

34
CHM136H1 Full Course Notes
Verified Note
34 documents
Document Summary
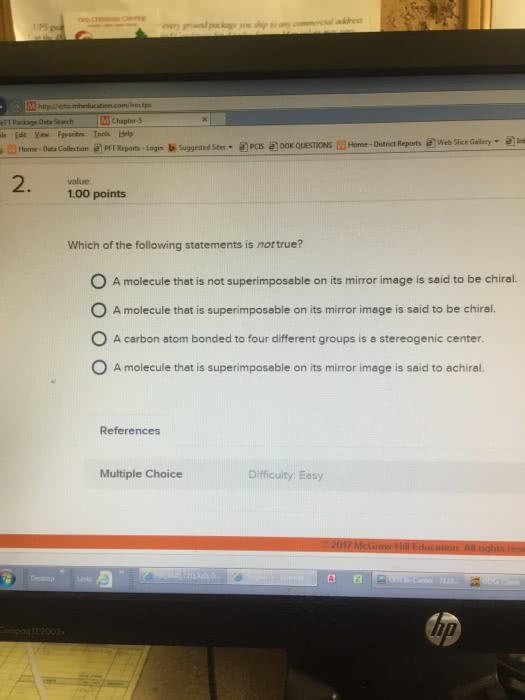
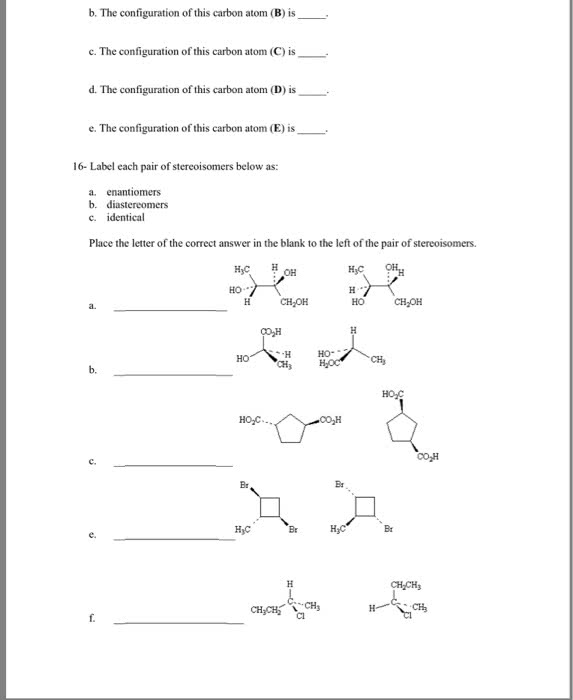
Handedness arises primarily as a consequence of the tetrahedral stereochemistry of sp3-hybridized carbon atoms. Molecules that aren"t identical to their mirror image are kinds of stereoisomers called enantiomers. A molecule not identical to its mirror image is said to be chiral. A molecule is not chiral if it has a plane of symmetry - it is achiral. Most common cause of chirality in an organic molecule is the presence of a tetrahedral carbon atom bonded to four different groups (chirality center/stereocenter/asymmetric center/stereogenic center) Chirality is a property of a molecule vs. the chiral center is the cause of chirality. Optically active organic molecules - plane of polarization rotated about the angle. Angle of rotation measured with a polarimeter (setup above - rotate analyzer until most light seen) Rotation left is given minus sign and rotation right is given a plus sign.