BCH210H1 Midterm: Study notes for midterm

49
BCH210H1 Full Course Notes
Verified Note
49 documents
Document Summary
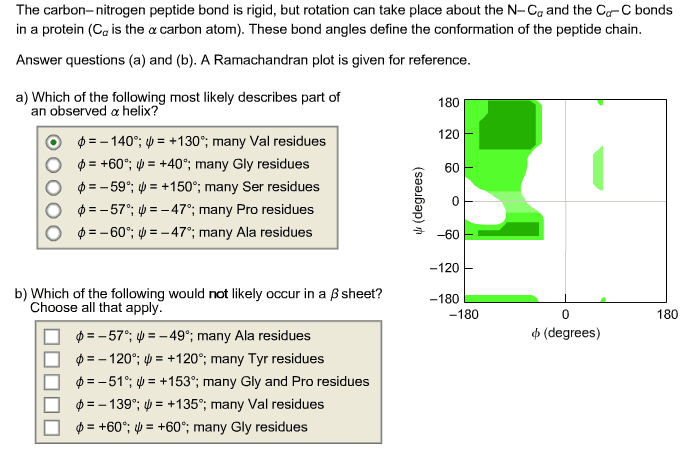
3 types phi = angle around the n-alpha c bond psi = angle around the alpha c-c=o bond omega = angle around the c-n bond chi angles reserved for side chain rotation. *** alpha helix: (phi, psi) = (-60 degrees, -50 degrees) *** beta sheet: (phi, psi) = (-140 degrees, +135 degrees) *** polyproline helix: (phi, psi) = (-60 degrees, +135 degrees) L-residues in proteins has 25% (phi, psi) space normally populated: ex. When stretched, intra-helix hydrogen bonds can be broken and inter-strand hydrogen bonds form via alpha-helix to beta-sheet transition sulfur present => disulfide crosslinks help restore to original shape. Resonance structures give c-n bond partial double-bond character. Peptide bond = resonance hybrid of two extremes, thus bond is planar: partial double-bond character, dihedral angle omega fixed, planar and trans. Inter-chain hydrogen bonds formed while side chains are above and below the plane of the hydrogen-bond network. Strong fibres that resist tension via covalent bonds also collagen.