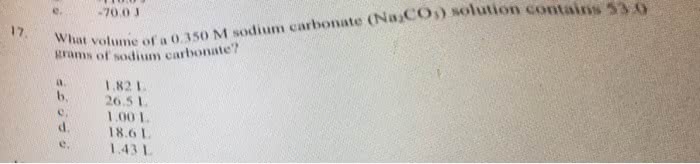j42s
J singhInternational Institute of Information Technology - IIIT Hyderabad
0 Followers
0 Following
0 Helped
j42sLv6
9 Sep 2022
Answer: Step-by-step explanation:13
j42sLv6
9 Sep 2022
Answer: 29Step-by-step explanation:Neutral copper has 29 protons which is same...
j42sLv6
9 Sep 2022
Answer: Step-by-step explanation:N3- contains 10 electrons and Na+ also contai...
j42sLv6
9 Sep 2022
Answer: 29Step-by-step explanation:Cu and Cu+ contains the same number of prot...
j42sLv6
9 Sep 2022
Answer: non polarStep-by-step explanation:As we know that dipole moment of CH4...
j42sLv6
9 Sep 2022
Answer: C-H bond is strongerStep-by-step explanation:We know that bond length ...
j42sLv6
9 Sep 2022
Answer: Step-by-step explanation:HCl+Zn ----> ZnCl2 +H2(gas)
j42sLv6
9 Sep 2022
Answer: Step-by-step explanation:MgSO4+BaCl2 -----> BaSO4+MgCl2 Note: BaSO4...
j42sLv6
9 Sep 2022
Answer: AgClStep-by-step explanation:AgCl (white precipitate) is formed on the...
j42sLv6
9 Sep 2022
Answer: F-,O2-,C4-Step-by-step explanation:N3- contains 7+3=10 electronsso F-,...
j42sLv6
9 Sep 2022
Answer: Step-by-step explanation:The equation derived is PM=dRT where P=pressu...
j42sLv6
9 Sep 2022
Answer: O2-,N3-, C4-Step-by-step explanation:F- contains 9+1= 10electrons Henc...
j42sLv6
9 Sep 2022
Answer: PV=nRTStep-by-step explanation:Gas equation derived is PV=nRT where P=...
j42sLv6
9 Sep 2022
Answer: 1.8066 *10^24Step-by-step explanation:In 1 molecule of h2so4 , it will...
j42sLv6
9 Sep 2022
Answer: 0.39112 molesStep-by-step explanation:Molar mass of CuBr2=223.5number ...
j42sLv6
9 Sep 2022
Answer: 6.67 %Step-by-step explanation:total mass of ch3cooh=60 mass of hydrog...
j42sLv6
9 Sep 2022
Answer: 76.19 %Step-by-step explanation:mass of h in nitric acid=1 mass of o i...
j42sLv6
9 Sep 2022
Answer: 28.4116 %Step-by-step explanation:mass of cu=63.5mass of cubr2= 63.5 +...
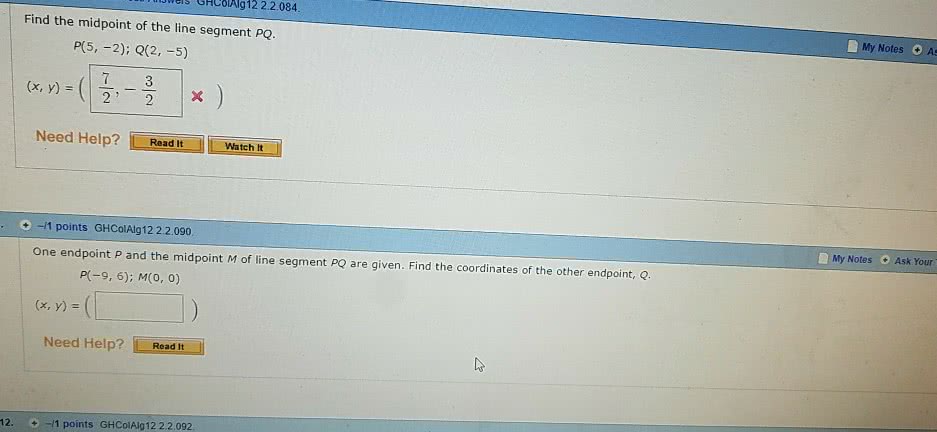
j42sLv6
8 Sep 2022
Answer: (7/2 ,-7/2)Step-by-step explanation:midpoint of two points (a,b) and (...
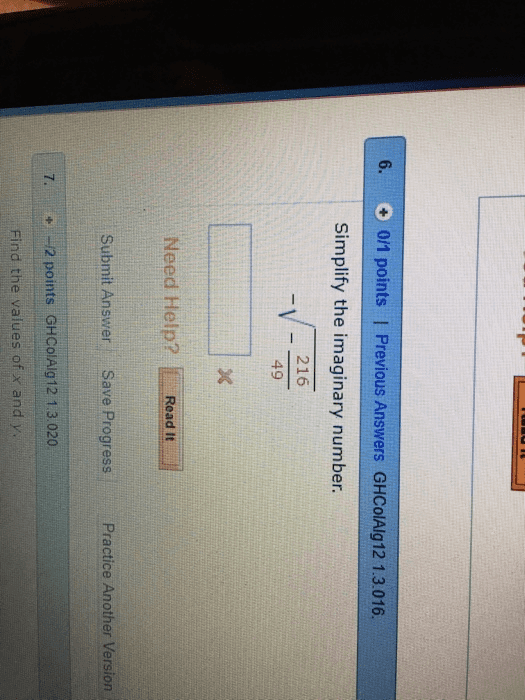
j42sLv6
7 Sep 2022
Answer: -6sqrt(6)/7 * iStep-by-step explanation:we know that sqrt(-1)= iso -sq...
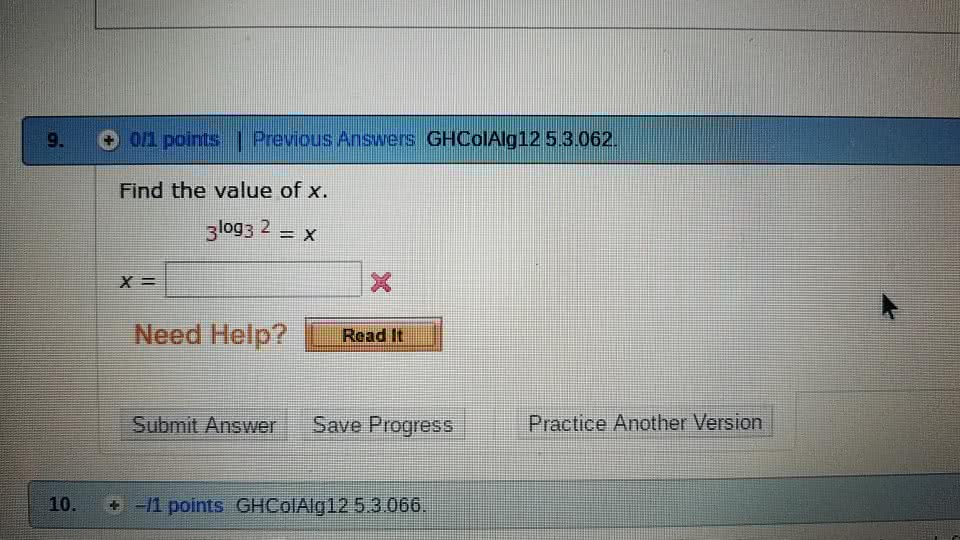
j42sLv6
7 Sep 2022
Answer: 2Step-by-step explanation:Using the property of log that a^(log(a) b) ...
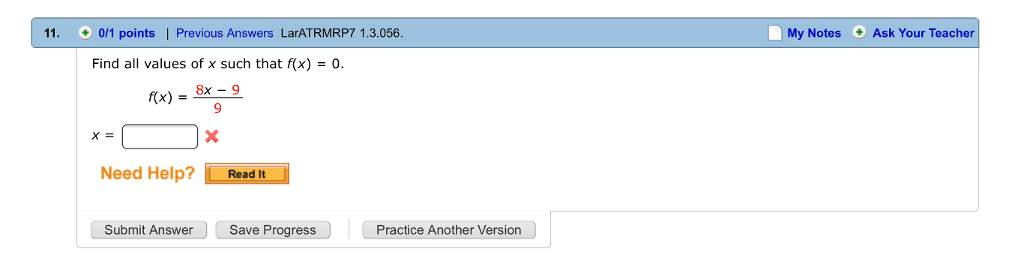
j42sLv6
7 Sep 2022
Answer: x=9/8Step-by-step explanation:f(X)=(8x-9)/9=08x-9=0x=9/8
j42sLv6
7 Sep 2022
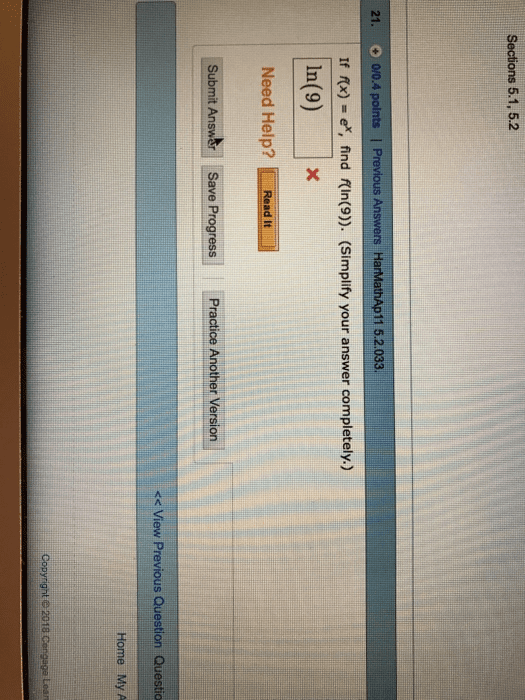
Answer: 9Step-by-step explanation:f(ln9)= e^(ln9)= e^(log (e) 9)= 9 (there is ...
j42sLv6
7 Sep 2022
Answer: x=158489.3192 approxStep-by-step explanation:log (10) x = 5.2x= 10^5.2...
j42sLv6
7 Sep 2022
Answer: 4Step-by-step explanation:log 10 (10^x)=4x log(10) 10 =4 (property of ...
j42sLv6
7 Sep 2022
Answer: 1Step-by-step explanation:log (10) 10 = 1
j42sLv6
7 Sep 2022
Answer: -zt+6t^2Step-by-step explanation: (2zt - z² + 5t²) + (z² - 3zt + t²) =...
j42sLv6
7 Sep 2022
Answer: x=2,3Step-by-step explanation:Assuming you want to find the values of ...
j42sLv6
7 Sep 2022
Answer: 0 Step-by-step explanation:Fe(CN)6 4- is a low spin complex as CN- is ...
j42sLv6
7 Sep 2022
Answer: Step-by-step explanation:SCN- is a weak field ligand and as we can see...
j42sLv6
7 Sep 2022
Answer: 70 , 0.7Step-by-step explanation:7/10 *100 = 70 % (percentage)decimal ...
j42sLv6
7 Sep 2022
Answer: 17/3Step-by-step explanation:3v-9=83v=17v=17/3
j42sLv6
7 Sep 2022
Answer: v=b-x+2Step-by-step explanation:x=b-v+2v=b-x+2
j42sLv6
7 Sep 2022
Answer: 12*11 =132Step-by-step explanation:All are correct except the last one...
j42sLv6
7 Sep 2022
Answer: 0.6MStep-by-step explanation:Molarity= (Number of moles of solute)/ vo...
j42sLv6
7 Sep 2022
Answer: volume=1.42857 LitersStep-by-step explanation:Molarity= (Number of mol...
j42sLv6
7 Sep 2022
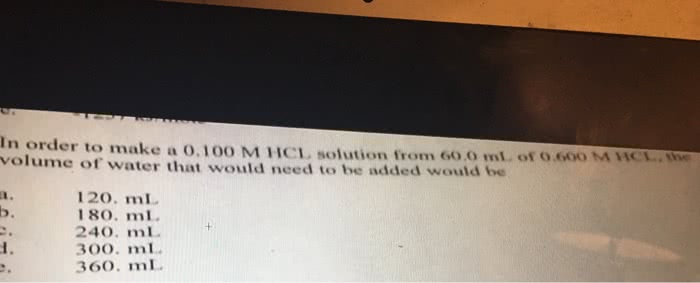
Answer: V1=360mlStep-by-step explanation:Here we will use the formula M1V1= M2...
j42sLv6
7 Sep 2022
Answer: 61.323642 LitersStep-by-step explanation:1 gallon =3.78541 Liters so 1...
j42sLv6
7 Sep 2022
Answer: EStep-by-step explanation:sin^2x=1/4sinx= +1/2 , -1/2 so x=pi/6,5pi/6,...
j42sLv6
7 Sep 2022
Answer: 10000Step-by-step explanation:I am assuming you are asking the value o...