2
answers
0
watching
197
views
12 Nov 2021
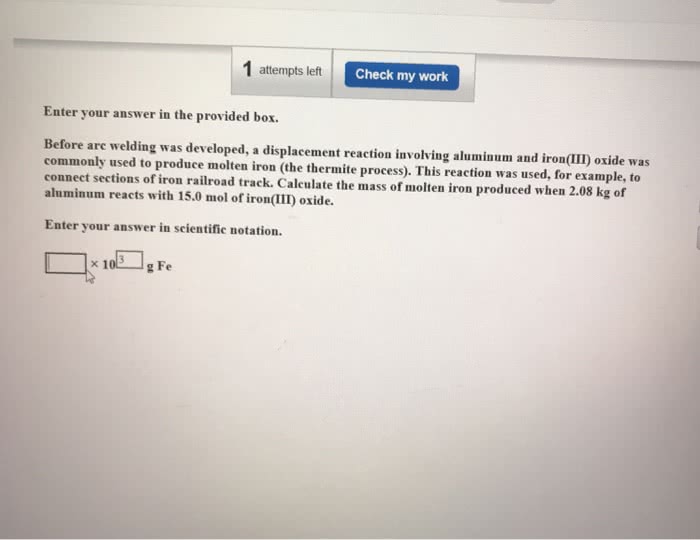
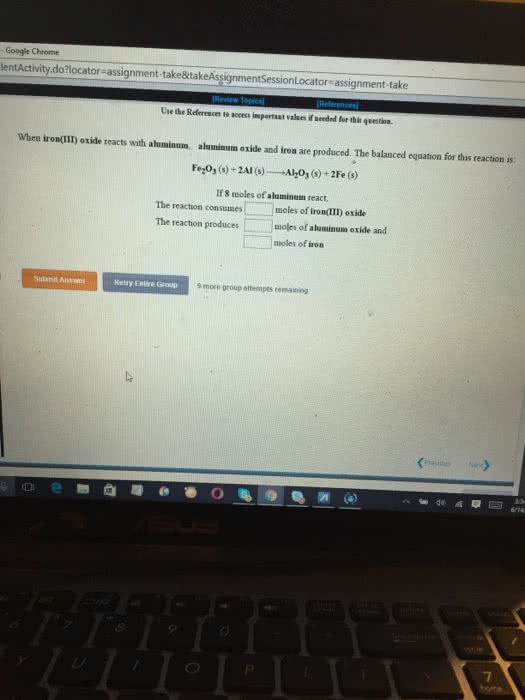
Thermite is a mixture of iron(III) oxide and aluminum powders that was once used to weld railroad tracks. It undergoes a spectacular reaction to yield solid aluminum oxide and molten iron. How many moles of iron(III) oxide are needed to form 3.603103 mol of iron?
Thermite is a mixture of iron(III) oxide and aluminum powders that was once used to weld railroad tracks. It undergoes a spectacular reaction to yield solid aluminum oxide and molten iron. How many moles of iron(III) oxide are needed to form 3.603103 mol of iron?
Read by 2 people
ahsanirshadLv1
12 Nov 2021
Read by 2 people
devrahycinthLv1
12 Nov 2021
Already have an account? Log in