1
answer
2
watching
632
views
15 Jan 2022
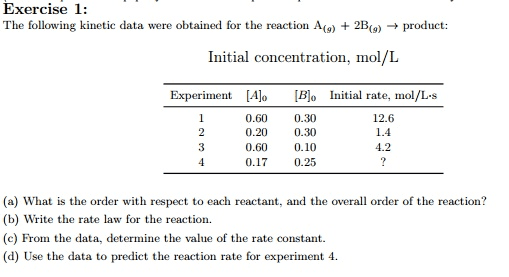
At a particular temperature and volume, two gases, A (red) and B (blue), react. The
following molecular scenes represent starting mixtures for four experiments:Expt no.: 1 2 3 4
Initial rate (mol/Ls): 0.000050 0.00010 0.00020 ?(a) What is the reaction order with respect to A, B and the overall order?
(b) Write the rate law for the reaction.
(c) Predict the initial rate of Expt 4.
At a particular temperature and volume, two gases, A (red) and B (blue), react. The
following molecular scenes represent starting mixtures for four experiments:Expt no.: 1 2 3 4
Initial rate (mol/Ls): 0.000050 0.00010 0.00020 ?(a) What is the reaction order with respect to A, B and the overall order?
(b) Write the rate law for the reaction.
(c) Predict the initial rate of Expt 4.
Liked by kumail.islamian and 1 others