13
answers
1
watching
186
views
cmall98Lv1
10 Jun 2023
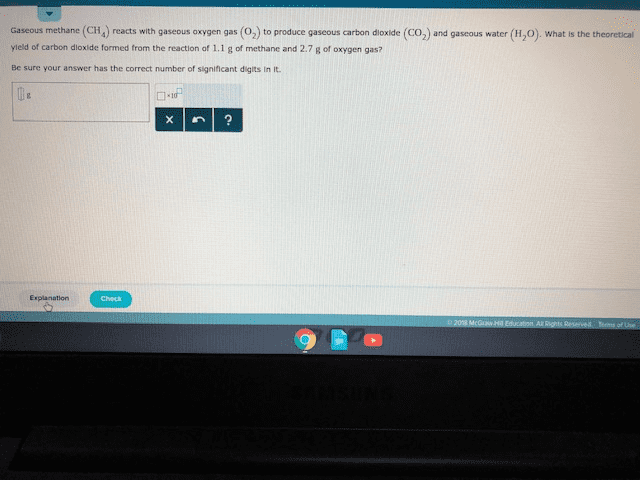
Gaseous methane (CH4) reacts with gaseous oxygen gas (O2) to produce gaseous carbon dioxide (CO2) and gaseous water (H2O). What is the theoretical yield of water formed from the reaction of 0.96g of methane and 3.3g of oxygen gas?
Round your answer to 2 significant figures
Gaseous methane (CH4) reacts with gaseous oxygen gas (O2) to produce gaseous carbon dioxide (CO2) and gaseous water (H2O). What is the theoretical yield of water formed from the reaction of 0.96g of methane and 3.3g of oxygen gas?
Round your answer to 2 significant figureskarimbalticLv7
3 Nov 2023
thanhmai1309Lv9
29 Aug 2023
Already have an account? Log in
bsuparna19Lv7
15 Aug 2023
Already have an account? Log in
wahabmunir796Lv10
11 Jul 2023
Already have an account? Log in
22 Jun 2023
Already have an account? Log in
13 Jun 2023
Already have an account? Log in
asifasabirLv10
12 Jun 2023
Already have an account? Log in
amirarsalanLv2
10 Jun 2023
Already have an account? Log in
10 Jun 2023
Already have an account? Log in
10 Jun 2023
Already have an account? Log in
Read by 1 person