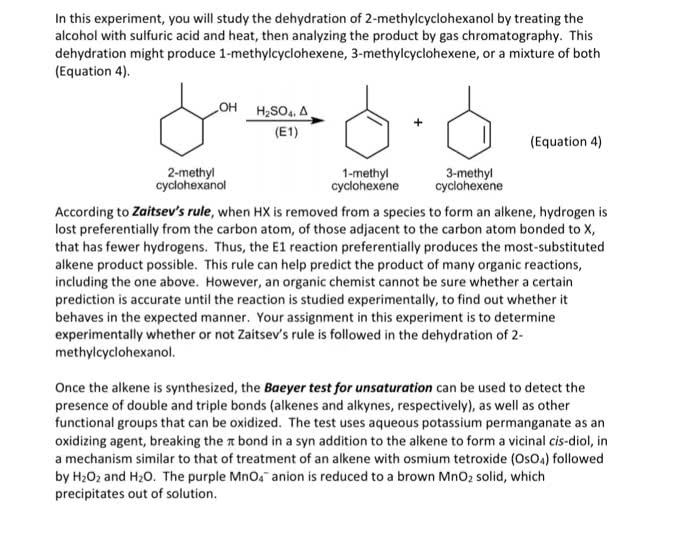
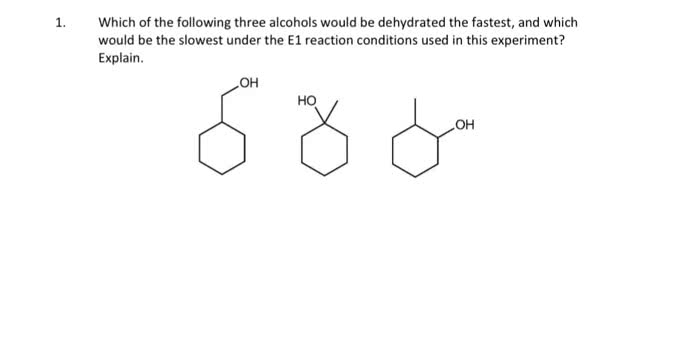
CHEM10009 Study Guide - Final Guide: Alkene, Adenine, Lone Pair

Alkene Addition Reactions
Proceed via a carbocation intermediate
-
Markovinkov's rule
Addition of H-X to an alkene, the halogen attaches itself to the more highly
substituted carbon atom
○
-
Mechanism:
-
Evidence for mechanism
Requires acid
○
Rate increases with acidity
○
Rate is independent of nucleophile concentration
○
Mixtures obtained if other nucleophiles present
○
-
Across periodic table, acidity increases with electronegativity
-
Down the periodic table, acidity increases with size
-
Unsymmetrical substrates (production of major and minor)
Carbocation stability
Stabilized by neighbouring carbon atoms
C-H bonds can donate some of e density to stabilise carbocation
○
Tertiary has more groups -> more stable
○
-
Stabilized by neighbouring C-C multiple bonds
Overlap b/w empty p orbital of carbocation with p orbitals of pi bond
allows charge to be shared b/w multiple atoms
○
-
Stabilized by adjacent lone pairs
Neighbouring can donate lone pair to e- poor carbocation -> formation of
double bond + charge moves to atom donating e- pair
○
-
Rearragnements when entire bonding pair of e- migrates to carbocation from
neighbour - favourable when new more stable carbocation formed
Hybride shifts = migrations of hydrogen with its lone pair
Alkyl shifts = migrations of a carbon atom with its lone pair
ADDITION POLYMERS
RADICAL POLYMERISATION???
Aromatic compounds
Molecules differ in chemical behaviour
-
More multiple bonds -> increasing energy required to hydrogenate
-
Benzene is more stable than predictions
-
Benzene
Hexagon
○
Resonance -> same bond lengths
○
Planar
○
Bond angles 120
○
Doesn’t behave like an alkene
○
Resonance stabilisation
Pi electrons are delocalised
§
○
REQUIREMENTS FOR AROMATICITY
Cyclic
-
Conjugated (alternating double and single bonds)
-
Planar
-
4n + 2 pi electrons (n must be an integer)
-
Heterocyclic aromatics
Heterocyclic = atoms of at least 2 elements in the ring
-
Lone pair in p orbital acts like a double bond -> aromatic
-
Pyridine
6 pi
○
N = 1 -> aromatic
○
-
Pyrrole
6 pi electrons
○
N = 1
○
-
Examples: adenine, guanine, thymine, cytosine (have resonance forms)
-
Disubstituted benzenes
Ortho
-
Meta
-
Para
-
Alkene Reactions
Tuesday, 29 May 2018
10:57 am

Alkene Addition Reactions
Proceed via a carbocation intermediate
-
Markovinkov's rule
Addition of H-X to an alkene, the halogen attaches itself to the more highly
substituted carbon atom
○
-
Mechanism:
-
Evidence for mechanism
Requires acid
○
Rate increases with acidity
○
Rate is independent of nucleophile concentration
○
Mixtures obtained if other nucleophiles present
○
-
Across periodic table, acidity increases with electronegativity
-
Down the periodic table, acidity increases with size
-
Unsymmetrical substrates (production of major and minor)
Carbocation stability
Stabilized by neighbouring carbon atoms
C-H bonds can donate some of e density to stabilise carbocation
○
Tertiary has more groups -> more stable
○
-
Stabilized by neighbouring C-C multiple bonds
Overlap b/w empty p orbital of carbocation with p orbitals of pi bond
allows charge to be shared b/w multiple atoms
○
-
Stabilized by adjacent lone pairs
Neighbouring can donate lone pair to e- poor carbocation -> formation of
double bond + charge moves to atom donating e- pair
○
-
Rearragnements when entire bonding pair of e- migrates to carbocation from
neighbour - favourable when new more stable carbocation formed
Hybride shifts = migrations of hydrogen with its lone pair
Alkyl shifts = migrations of a carbon atom with its lone pair
ADDITION POLYMERS
RADICAL POLYMERISATION???
Aromatic compounds
Molecules differ in chemical behaviour
-
More multiple bonds -> increasing energy required to hydrogenate
-
Benzene is more stable than predictions
-
Benzene
Hexagon
○
Resonance -> same bond lengths
○
Planar
○
Bond angles 120
○
Doesn’t behave like an alkene
○
Resonance stabilisation
Pi electrons are delocalised
§
○
REQUIREMENTS FOR AROMATICITY
Cyclic
-
Conjugated (alternating double and single bonds)
-
Planar
-
4n + 2 pi electrons (n must be an integer)
-
Heterocyclic aromatics
Heterocyclic = atoms of at least 2 elements in the ring
-
Lone pair in p orbital acts like a double bond -> aromatic
-
Pyridine
6 pi
○
N = 1 -> aromatic
○
-
Pyrrole
6 pi electrons
○
N = 1
○
-
Examples: adenine, guanine, thymine, cytosine (have resonance forms)
-
Disubstituted benzenes
Ortho
-
Meta
-
Para
-
Alkene Reactions
Tuesday, 29 May 2018
10:57 am

Alkene Addition Reactions
Proceed via a carbocation intermediate
-
Markovinkov's rule
Addition of H-X to an alkene, the halogen attaches itself to the more highly
substituted carbon atom
○
-
Mechanism:
-
Evidence for mechanism
Requires acid
○
Rate increases with acidity
○
Rate is independent of nucleophile concentration
○
Mixtures obtained if other nucleophiles present
○
-
Across periodic table, acidity increases with electronegativity
-
Down the periodic table, acidity increases with size
-
Unsymmetrical substrates (production of major and minor)
Carbocation stability
Stabilized by neighbouring carbon atoms
C-H bonds can donate some of e density to stabilise carbocation
○
Tertiary has more groups -> more stable
○
-
Stabilized by neighbouring C-C multiple bonds
Overlap b/w empty p orbital of carbocation with p orbitals of pi bond
allows charge to be shared b/w multiple atoms
○
-
Stabilized by adjacent lone pairs
Neighbouring can donate lone pair to e- poor carbocation -> formation of
double bond + charge moves to atom donating e- pair
○
-
Rearragnements when entire bonding pair of e- migrates to carbocation from
neighbour - favourable when new more stable carbocation formed
Hybride shifts = migrations of hydrogen with its lone pair
Alkyl shifts = migrations of a carbon atom with its lone pair
ADDITION POLYMERS
RADICAL POLYMERISATION???
Aromatic compounds
Molecules differ in chemical behaviour
-
More multiple bonds -> increasing energy required to hydrogenate
-
Benzene is more stable than predictions
-
Benzene
Hexagon
○
Resonance -> same bond lengths
○
Planar
○
Bond angles 120
○
Doesn’t behave like an alkene
○
Resonance stabilisation
Pi electrons are delocalised
§
○
REQUIREMENTS FOR AROMATICITY
Cyclic
-
Conjugated (alternating double and single bonds)
-
Planar
-
4n + 2 pi electrons (n must be an integer)
-
Heterocyclic aromatics
Heterocyclic = atoms of at least 2 elements in the ring
-
Lone pair in p orbital acts like a double bond -> aromatic
-
Pyridine
6 pi
○
N = 1 -> aromatic
○
-
Pyrrole
6 pi electrons
○
N = 1
○
-
Examples: adenine, guanine, thymine, cytosine (have resonance forms)
-
Disubstituted benzenes
Ortho
-
Meta
-
Para
-
Alkene Reactions
Tuesday, 29 May 2018 10:57 am
Document Summary
Addition of h-x to an alkene, the halogen attaches itself to the more highly substituted carbon atom. Down the periodic table, acidity increases with size. C-h bonds can donate some of e density to stabilise carbocation. Overlap b/w empty p orbital of carbocation with p orbitals of pi bond allows charge to be shared b/w multiple atoms. Neighbouring can donate lone pair to e- poor carbocation -> formation of double bond + charge moves to atom donating e- pair. Rearragnements when entire bonding pair of e- migrates to carbocation from neighbour - favourable when new more stable carbocation formed. Hybride shifts = migrations of hydrogen with its lone pair. Alkyl shifts = migrations of a carbon atom with its lone pair. More multiple bonds -> increasing energy required to hydrogenate. 4n + 2 pi electrons (n must be an integer) Heterocyclic = atoms of at least 2 elements in the ring.