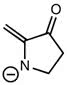
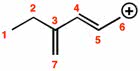
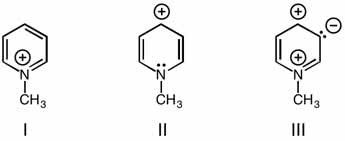
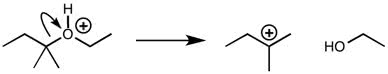
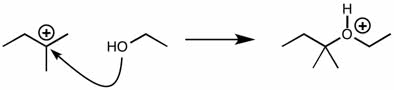
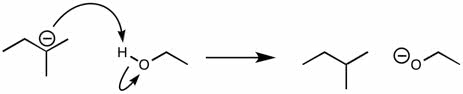
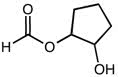
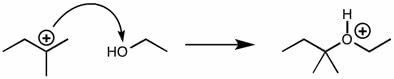CHMI-2426EL Study Guide - Quiz Guide: Newman Projection, Acid Dissociation Constant, Cyclohexane
Document Summary
Covalent: sharing electrons in space: sharing electrons between 2 atoms, overlapping of atomic orbitals. The mathematical function that describes the high probability of finding an electron around the nucleus. Forms only the single bond ( covalent bond) Covalent bond can be formed by sideways overlap between 2py or. Covalent bond can also be formed by linear (horizontal) overlap of. S-s and p-p overlaps lead to covalent bonds. Formed by s-s, s-px, and px-px (linear) Formed by py-py and pz-pz (sideways) Bond is much stronger than the bond ( is more reactive) Carbon z=6; need to determine shape of carbon atom within a molecule. S and p orbitals from same atom combine combine to form new hybrid atomic orbitals with high electron density. P orbital will form a bond. P orbitals will form 2 bonds. Sharing electrons unequally polar (dipole moment = ) Electronegativities can be used to predict whether a bond will be polar.