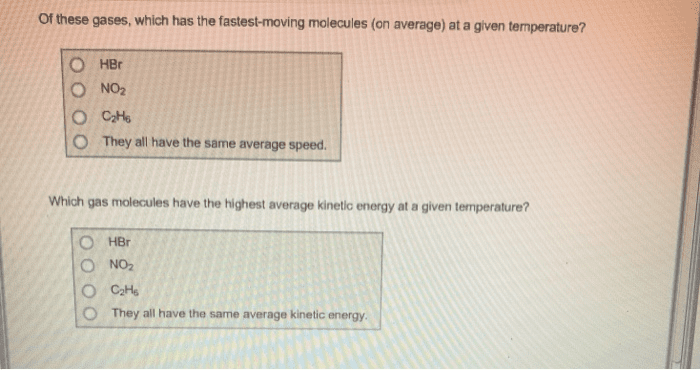
of these gases, which has the fastest-moving molecules (on average) at a given temperature? O HBr O NO2 O They all have the same average speed Which gas molecules have the highest average kinetic energy at a given temperature? O NO2 O They all have the same average kinetic energy
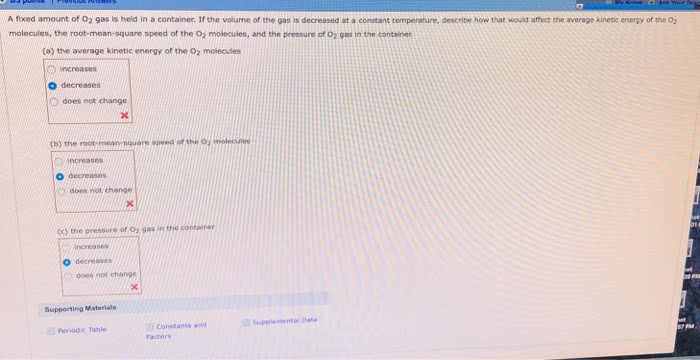
Consider equimolar samples of different ideal gases at the same volume and temperature. Gas A has a higher molar mass than gas B. Compare the pressures. Compare the rms speeds. Compare the average kinetic energies Consider equimolar samples of the same ideal gas at the same volume, but different temperatures. Sample C is at a higher temperature than sample D Compare the pressures. Compare the rms speeds. Compare the average kinetic energies. Consider equimolar samples of the same ideal gas at the same termperature, but different volumes Sample E has a larger volume than sample F pare the pressures Compare the rms speeds.Compare the average kinetic energies.
Container A holds 772 mL of ideal gas at 2.80 atm. Container B holds 164 mL of ideal gas at 4.60 atm. If the gases are allowed to mix together, what is the resulting pressure? Number atm
Show transcribed image text of these gases, which has the fastest-moving molecules (on average) at a given temperature? O HBr O NO2 O They all have the same average speed Which gas molecules have the highest average kinetic energy at a given temperature? O NO2 O They all have the same average kinetic energy
Consider equimolar samples of different ideal gases at the same volume and temperature. Gas A has a higher molar mass than gas B. Compare the pressures. Compare the rms speeds. Compare the average kinetic energies Consider equimolar samples of the same ideal gas at the same volume, but different temperatures. Sample C is at a higher temperature than sample D Compare the pressures. Compare the rms speeds. Compare the average kinetic energies. Consider equimolar samples of the same ideal gas at the same termperature, but different volumes Sample E has a larger volume than sample F pare the pressures Compare the rms speeds.Compare the average kinetic energies.
Container A holds 772 mL of ideal gas at 2.80 atm. Container B holds 164 mL of ideal gas at 4.60 atm. If the gases are allowed to mix together, what is the resulting pressure? Number atm