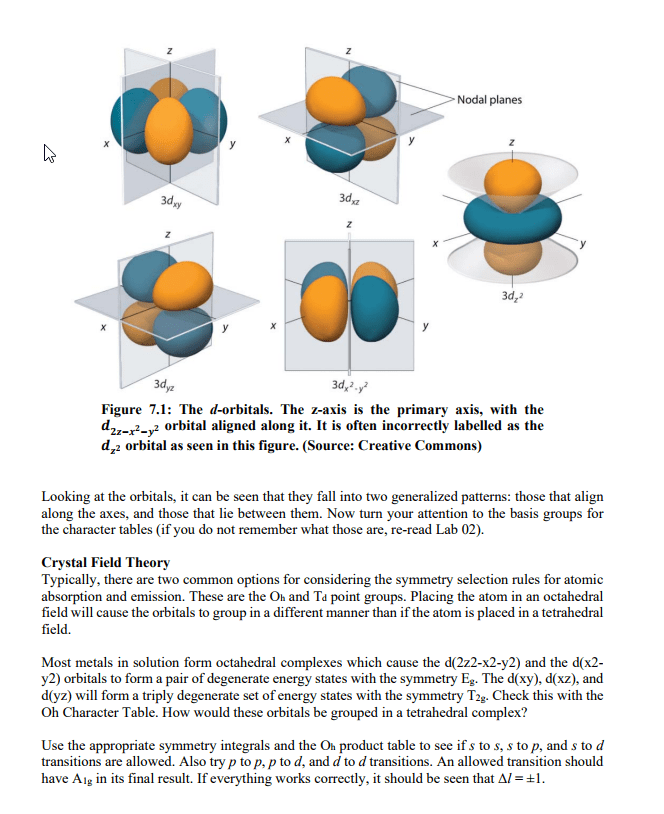
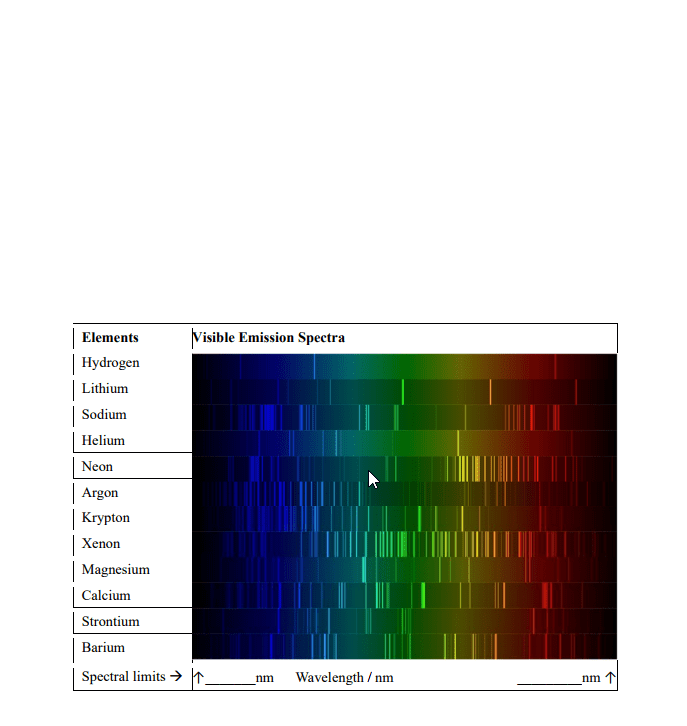
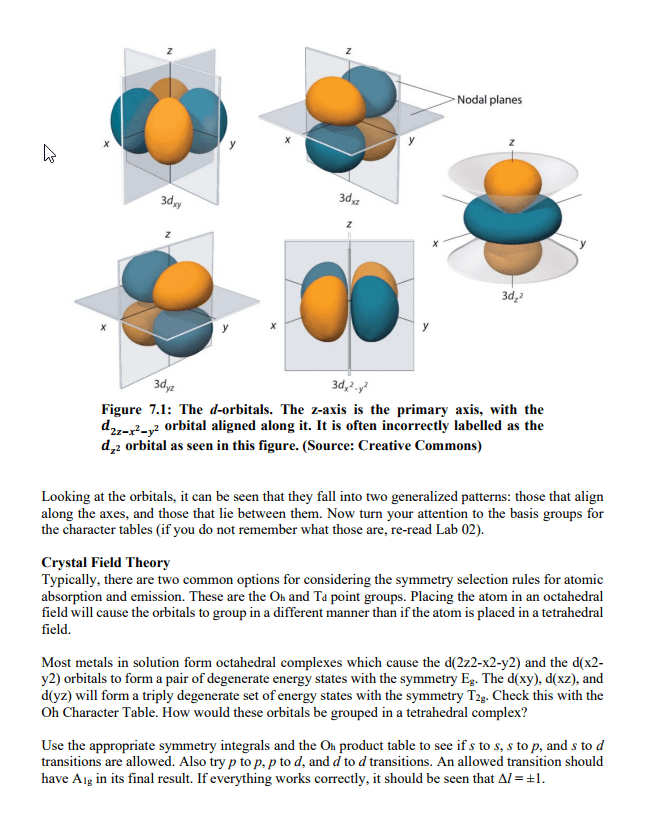
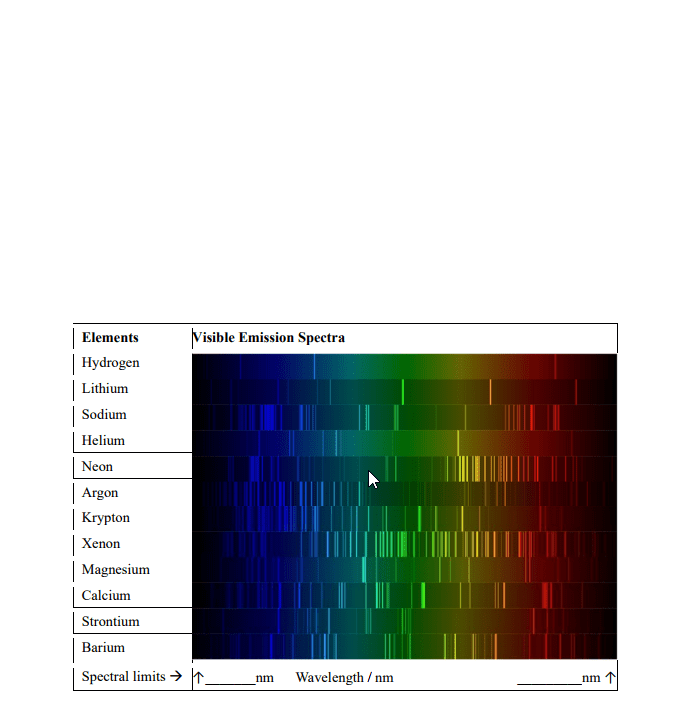
CHEM 1A03 Study Guide - Final Guide: Emission Spectrum, Diamagnetism, Paramagnetism
60 views5 pages
12 Dec 2017
School
Department
Course
Professor

35
CHEM 1A03 Full Course Notes
Verified Note
35 documents
Document Summary
As electrons relax back down, each energy level difference is discrete or quantized, and corresponds to a single wavelength. All these discrete wavelengths would be mixed into one colour, but if we allow this light to pass through a narrow slit and through a prism, we can separate out all of the different wavelengths (color) We would get an atomic emission spectrum. Atomic absorption spectrum, we see all wavelengths of light except those that are being absorbed, which appear as black bands in the spectrum. When you collect the emission and absorption spectrum for the same sample, the lines correspond to the gaps. Not limited to just atoms but also apply to molecules. Allows us to identify presence of different molecules in samples since each atom or molecules has a unit set of bands. 9. 109 x 10^-31 kg = mass of electron (m) Mass (m) x speed (u) also equals momentum (p)
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232