CHEM101 Study Guide - Dan Piraro, Molecular Electronic Transition, Bohr Model

77
CHEM101 Full Course Notes
Verified Note
77 documents
Document Summary
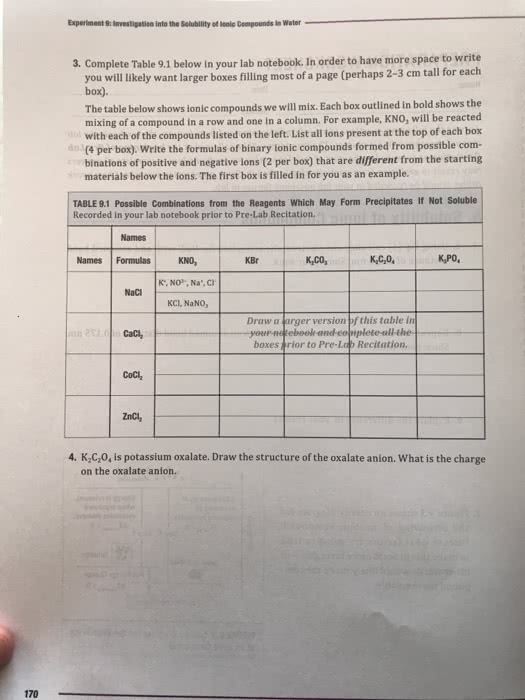
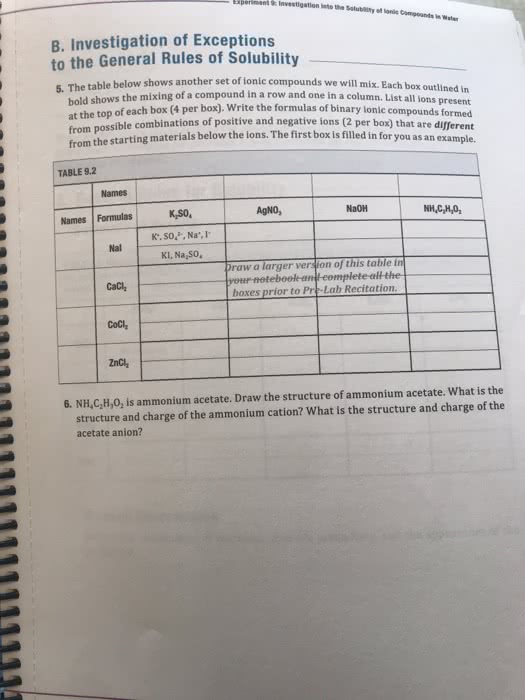
Read textbook sections 7. 4, 8. 1 can solve questions 1 4. 1st online lecture is next week (likely w 2pm f 6am). 1 e orbiting around a positive nucleus (really ions) He 2 e , 2 p+ not conforming to the model. It must lose 1 e to be like h so that the bohr equation can be used. Li 3 e , 3 p+ not conforming to the model. It must lose 2 e to be like h so that the bohr equation can be used. The figure below represents the emission spectrum of a one electron ion in the gas phase. All lines result from transitions to the ground state. Line a has a wavelength of 6. 1 nm. (a) what electronic transition corresponds to line a? (b) identify the ion that exhibits the spectrum. Each line has 2 n values associated with it, ni and nf !