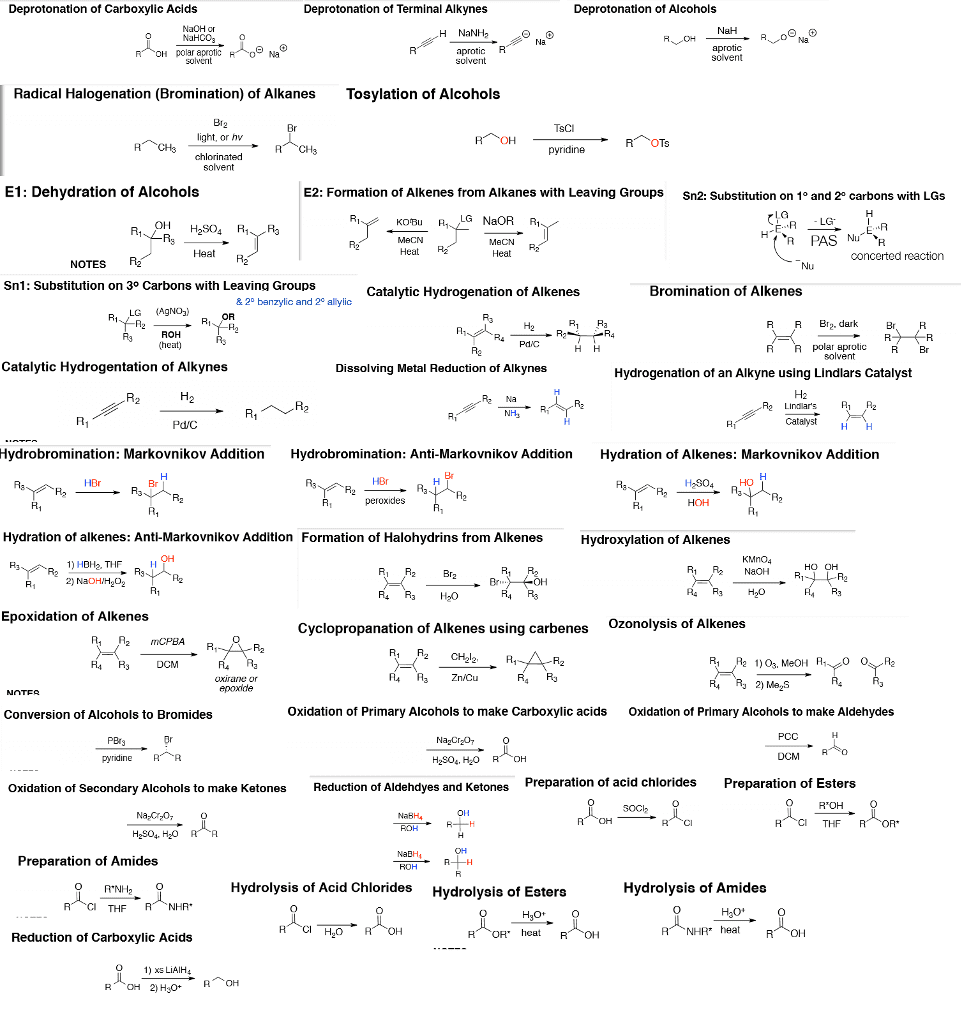
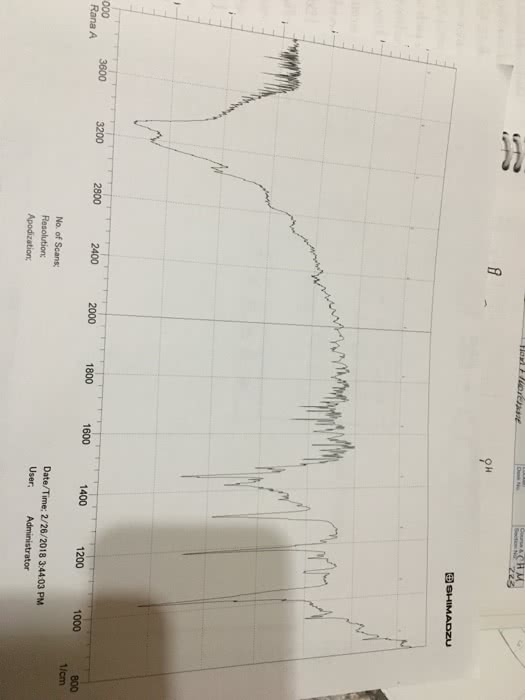
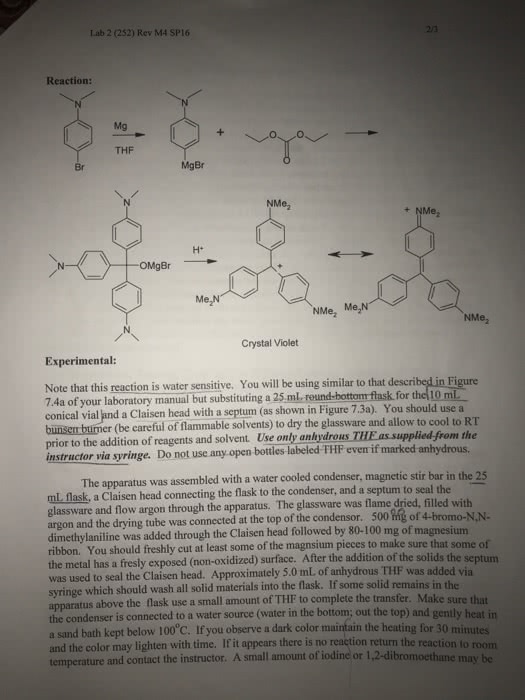
Total amount of dye obtained (crystal violet) = 6.5mL
The absorbance using UV spectrometer is =1.75
Please help within the following
Using the reported value of the extinction coefficient of 112,000 M^-1 cm^-1, calculate the concentration of diluted sample and (according for dilution ) estimate the percent yield of this reaction by determining the number of moles of the dye formed. You can assume that the dye follows the Beer-Lambert Law and that A=ebc which for a 1cm cell simplifies to Absorbance A= ( 112,000 M^-1 cm^-1)(1.00cm) (concentration of crystal violet).
Please help thank you!
Organic Laborat 2. The Grignard Reaction: Preparation of Crystal Violet Reference: Journal of Chemical Education, 73, 259, (1996) Douglass Taber, Robert Meagley and Danielle Supplee Laboratory Objectives: 1. Prepare a Grignard Reagent from a bromoaromatic compound and magnesium. 2. Perform a water sensitive reaction at a micro-scale. 3. Observe the visible color of a highly conjugated triphenylmethyl dye. 4. Predict reaction products expected from the reaction of Grignard Reagents with carbonyl compounds. Introduction: The Grignard Reaction is a chemical reaction which to form the Grignard reagent would be classified as a reduction. Magnesium reacts with alkyl and aryl halide to form an organometallic reagent which can subsequently react as a carbon nucleophile in a chemical reaction. Grignard reagents can be stored as THF solutions but it is common to make the organometallic compound and then react it without work-up with some suitable compound. This could be carbon dioxide to form a carboxylic acid or water or deuterium oxide to form the reduced hydrocarbon. More commonly carbonyl compounds (organic compounds which contain a C-O group) are used to form the addition product of the organometallic compound and the carbonyl group. This results in an alcohol if aldehydes and ketones are used as the carbonyl group. If the carbonyl group used is an ester (R-COp-R') then the nucleophilic addition to the carbonyl carbon occurs as expected but the carbonyl is reformed by loss of the alkoxide ion. This new carbonyl compound can add an additional mole of organometallic reagent to again give an alcohol but one that contains two of the organic groups from the Grignard reagent. In this reaction you will prepare an aromatic organometallic reagent which will react with diethyl carbonate. Diethyl carbonate will perform the reaction described for esters above but since there are two alkoxide groups that can leave, this results in the addition of not two but three moles of the Grignard reagent. Also the product expected would be a tertiary alcohol in which each of the carbon groups is aromatic. The triphenyl methyl group is commonly called a "trityl" group and since the central carbon is both tertiary and benzylic to each of the rings the alcohol that might be expected will quickly dehydrate to give a stable carbocation (a so-called trityl or triphenylmethyl cation). In the compound that you will prepare called Crystal Violet or Gentian Violet the cation is not only a trityl cation but each of the rings has a dimethylamino group in the para position. These groups being electron donating by resonance will also help to stabilize the cation. This highly conjugated compound will absorb visible radiation and will be highly colored. This compound has been used as a dye in commercial applications including biological staining.