CHM 2123 Study Guide - Leaving Group, Alpha And Beta Carbon, Electrophile
Document Summary
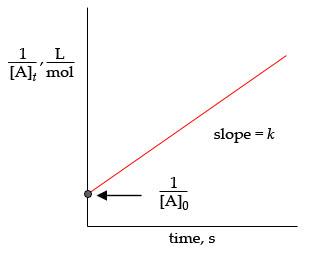
The purpose of this experiment is to study the kinetics of a reaction. It is important to understand the behaviour of molecules during the steps of a reaction. The zero order reaction is one that is zero order in respect with one reactant and first order in respect to the other, producing a linear graph with slope -k. The first order reaction is one that would depend on the concentration of one reactant. The rate law is expressed as: d[a]/dt=-kt + ln([a]o) First order reactions would produce a straight line graph of ln[a]t vs t with slope -k. Second order reactions depends on the concentration of both reactants, expressed by the equation: d[a]/dt=-kt + 1/[a]o. A second order reaction would produce a straight line graph of 1/[a]t vs t with slope k. In this lab substitution reactions will be observed. Substitution reactions are the reaction between nucleophiles (bearing a long pair) and electrophiles (empty orbital to accept lone pair).