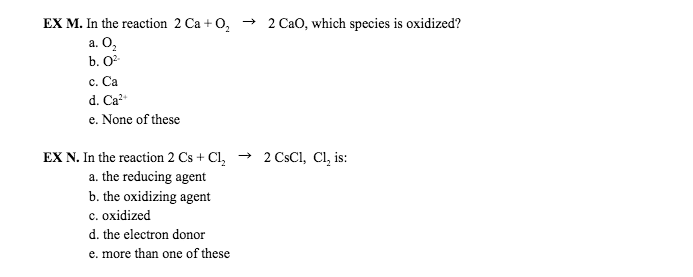CH110 Study Guide - Binary Phase, Molar Mass, Scientific Notation
Document Summary
Al2s3 (s) + h2o (l) al(oh)3 + h2s(g) Iib) write a balanced chemical equation for each of the following:(8) Iiia) write the chemical formula for each of the following(5) C3h8 (g) + 5o2 (g) 3 co2 (g) + 4h2o (g: 5. 54 x 1023 molecules, 1. 39 x 1023 molecules, 20. 3 x 1023 molecules, 2. 77 x 1023 molecules, 3. 89 x 1023 molecules. When this reaction is carried out in a given experiment, only 30g is produced. Calculate the empirical formula of this compound: cocl, cocl2, co2cl, co2cl2, cocl4. Calculate the empirical and the molecular formulas: c2h3 and c4h6, ch and c3h3, ch2 and c3h6, ch3 and c2h6, c2h6 and c3h9. The correct formula for arsenic(iii) oxide is: as3o3, as2o3, as3o2, aso, as3o. None of the above are correct: the sum of the coefficients when the following equation is balanced is. Mg3n2 (s) + h2o (l) nh3(g) + mg(oh)2 (s) Fecl3(aq) + koh (aq) fe (oh)3 + kcl (aq)