CHEM 1000 Study Guide - Final Guide: Gas Thermometer, Nitride, Xylene
157 views18 pages
12 Dec 2014
School
Department
Course
Professor
Document Summary
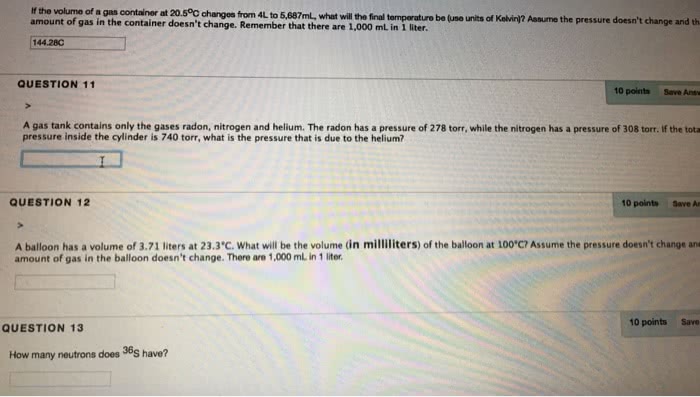
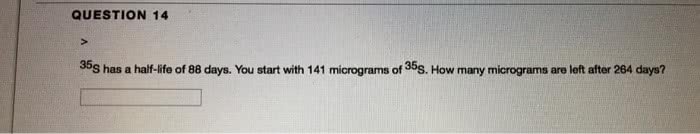
A gas thermometer can be used to determine temperature by measuring the pressure of a known amount of gas held in a bulb: a 6. 00 l gas bulb contains 6. 40g of oxygen (o2) at 23. 6 c. A small piece of lithium was placed in a 1. 6l container of pure nitrogen at a pressure of 1150 torr and 25. 0 c. After all the lithium had reacted the pressure in the container was reduced to 956 torr. What mass of lithium nitride has been produced: at what temperature is the average speed of nitrogen molecule twice that of an argon atom at 300k. 2: a flask contains twice as many molecules of argon as xylene (c8h10). Gas is allowed to escape through a small orifice in the flask. After 100s, 0. 010 mole of argon has escaped. Assume this heat is lost by the evaporation of sweat (water).
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232