CHM 101 Midterm: 101AExam2
168 views14 pages
Document Summary
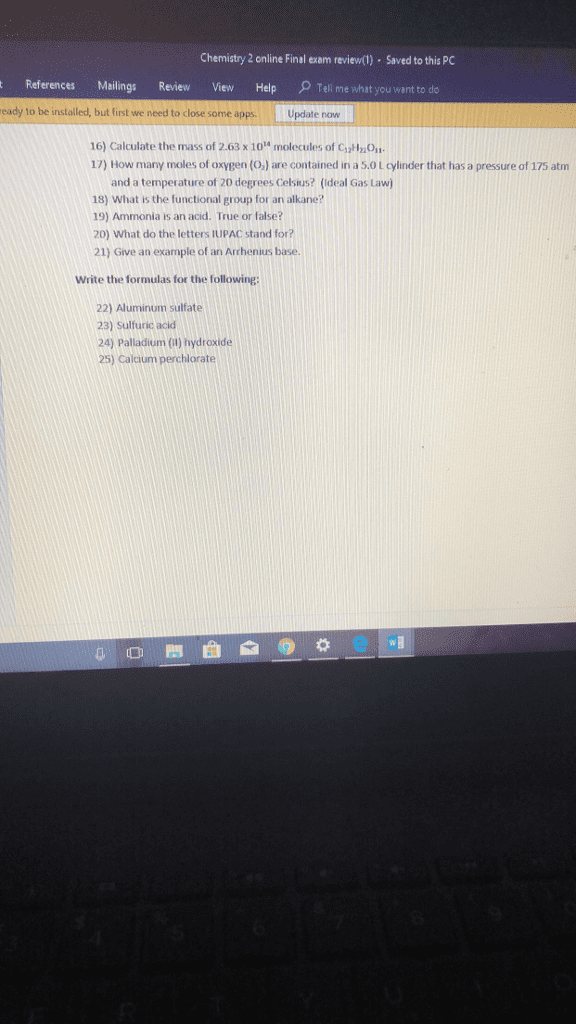
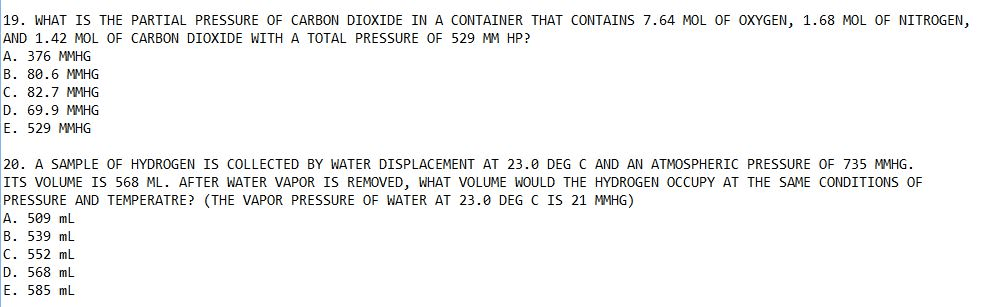
Version 1: given the following molecular formulas, determine the empirical formula of each compound: n2o5, h2o2, c6h4cl2, n2o5, h2o2, c3h2cl2, n2o5, ho, c3h2cl, n2o5, h2o, c6h4cl2, n2o5, ho, c6h4cl2, no2. 5, ho, c3h2cl. Acetic acid is the active ingredient in vinegar. Add another hydrogen atom to each molecule on the right: there are not enough hydrogen atoms in either of the two images. Add one more hydrogen molecule to the left image, and one more hydrogen atom to each molecule shown on the right: there are too few hydrogen atoms in the image on the left. Version 1: balance the following skeletal equation: c3h8(g) + o2(g) co2(g) + h2o(g), c3h8(g) + o2(g) 3co2(g) + 4h2o(g, c3h8(g) + 5o2(g) 3co2(g) + 4h2o(g) The class of this reaction is: combination reaction, single-displacement reaction, double-displacement reaction, decomposition reaction, combustion reaction, the class of the reaction shown in the figure is: Cu(s) + zn2no3(aq) cuno3(aq) + 2zn(aq: no reaction will occur, yes.