CHEM1161 Study Guide - Atomic Radius, Valence Electron, Atomic Orbital
Document Summary
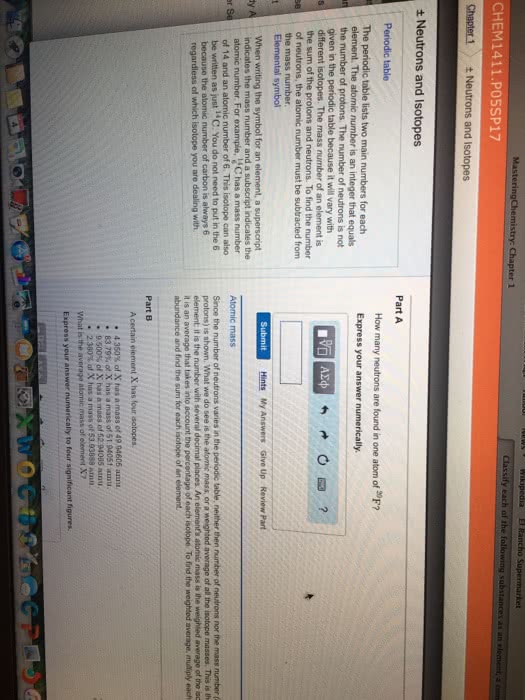
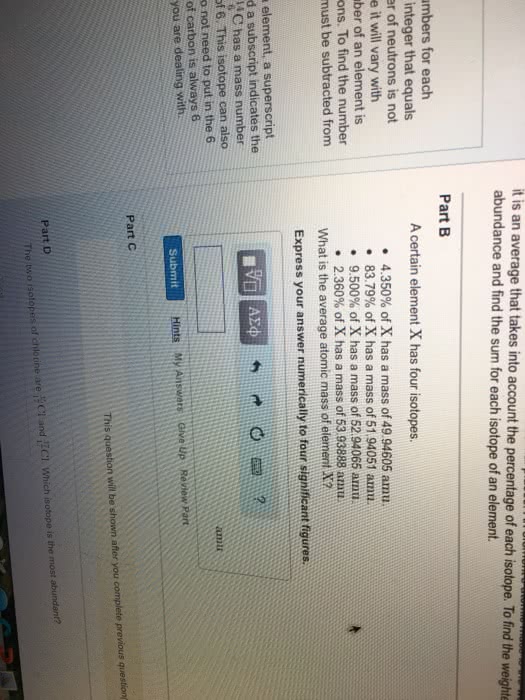
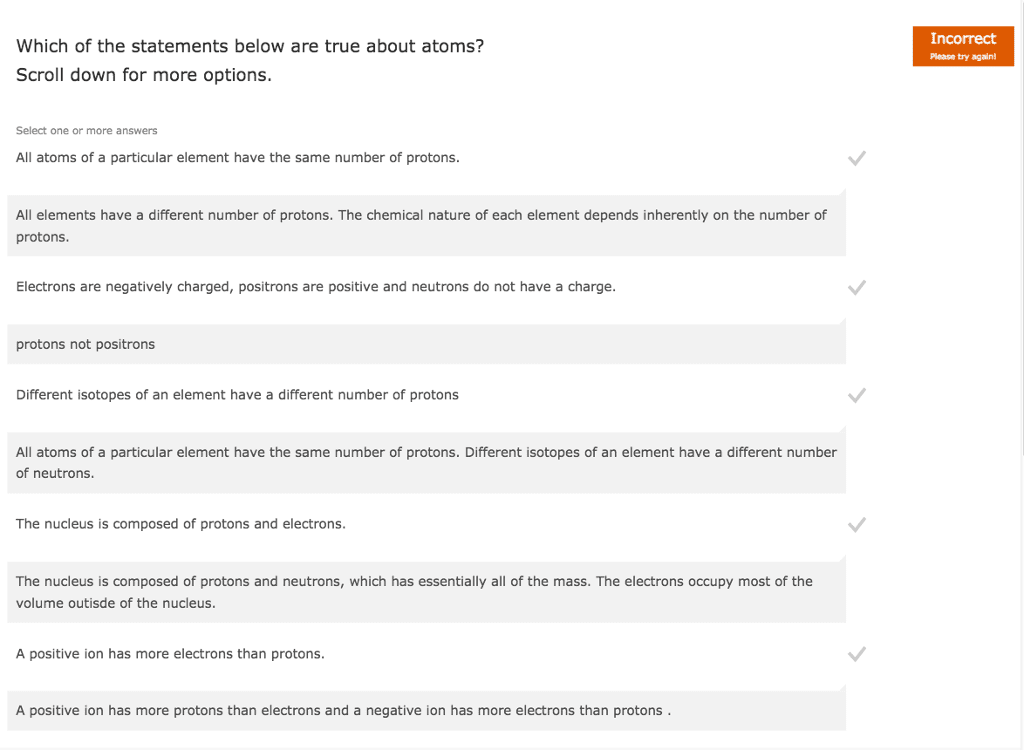
Neutral atoms: net charge is zero, number of protons=number of electrons. Mass number: represents the number of particles in the nucleus, # of protons +# of neutrons, always a whole number, does not appear in periodic table, number of neurons = mass number - atomic number. Electromagnetic spectrum: shows the arrangement of wavelengths of electromagnetic radiation within the visible range from 700 to 400 nm. E= hc/ h= constant plank"s constant v=frequency of wave c=speed of light lamda=wavlength: elements have unique atomic spectrum. Valence electrons: number of electrons in the outermost energy level, group number gives the number of valence electrons for the representative elements, valence electrons =group number for representative elements. Electron dot structures: lewis dot, represent valence electrons as dots placed on side of symbol al. Ionization energy: energy required to remove one of the outermost electrons from an atom. Decreases down a group, increases across a period from left to right.