CHEM 1315 Study Guide - Final Guide: Sodium Acetate, Buffer Solution, Quadratic Equation
188 views28 pages
10 May 2017
School
Department
Course
Professor
Document Summary
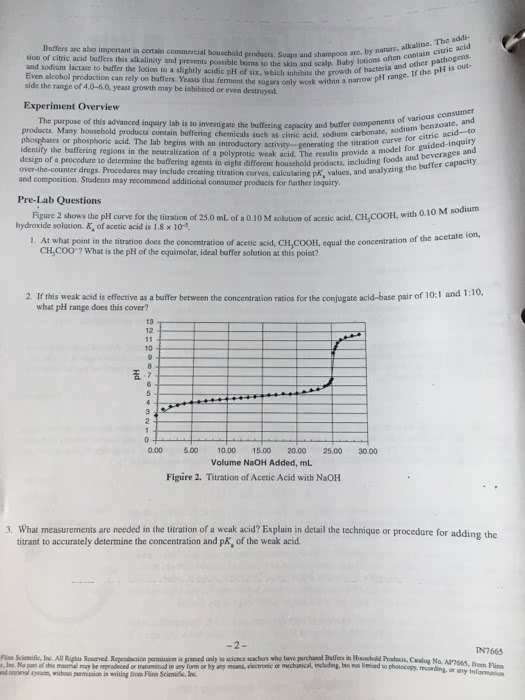
If we have two solutes each containing the same ion, the equilibrium is affected. Consider adding acetic acid (ch3cooh) and sodium acetate (nach3coo) to water. The acetate anion from sodium acetate will inhibit acetic acid from dissociating (le chatelier). First consider the situation with only acetic acid, at 0. 10 m. X = 1. 34 10 3 which means the ph = 2. 87 and the % ionization = 1. 34% Next, consider adding 0. 050 moles of sodium acetate to 1. 00 l of our acetic acid solution (assume volume does not change). Equilibrium (note: the equilibrium must shift to the left from le chatelier) 1. 34 x 10 3 - x 5. 134 x 10 2 -x. This gives [h+] = 3. 5 x 10 5 (using the quadratic equation) which means the ph = 4. 46 and the % ionization = 0. 035% Comment: the last calculation was hard because we had to use the quadratic equation.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232