CHEM 120 Chapter Notes - Chapter 17: Sulfur Trioxide, Hemoglobin, Phenolphthalein

26
CHEM 120 Full Course Notes
Verified Note
26 documents
Document Summary
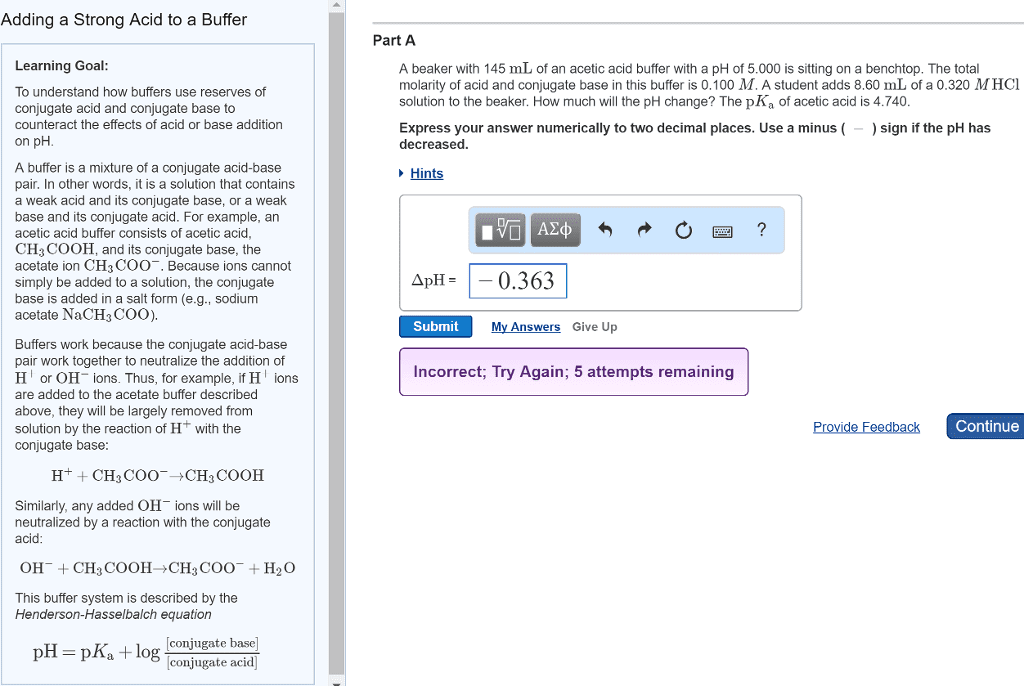
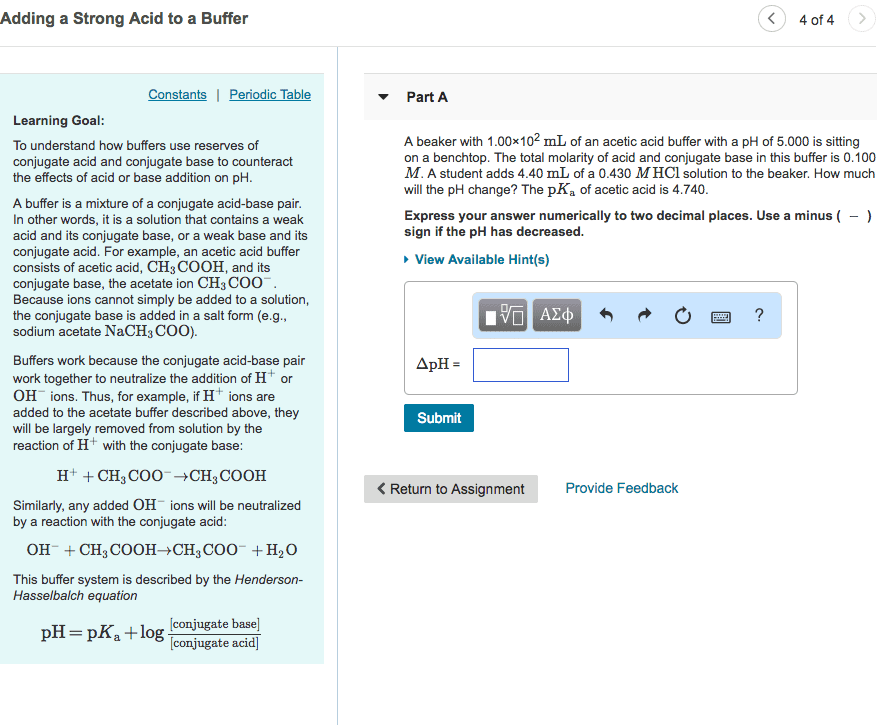
17. 1 equilibria of acid-base buffers: acid-base buffer: solution that lessens the impact on ph from the addition of acid or base. [h3o+] increase very slightly: strong base: addition of small amount of strong base produces opposite result. Buffer range: buffer range: ph range over which buffer is effective, related to relative buffer-component concentrations. Preparing a buffer: steps required to prepare a buffer, choose conjugate acid-base pair: deciding chemical composition based on desired ph. If want buffer ph 3. 90, pka of acid component close to 3. 90 (ka 10-3. 90) and choose. Hcooh and naoh: as oh- reacts with hcooh, neutralization of some of hcooh produces the hcoo- needed, hcooh (ha total) + oh- hcooh + hcoo- + h2o. Calculating ph during titration: calculate ph at various points by knowing chemical species present, before equivalence point: as start adding base, some acid neutralized and volume increases. Initial solution of strong ha: ph = -log[h3o+]