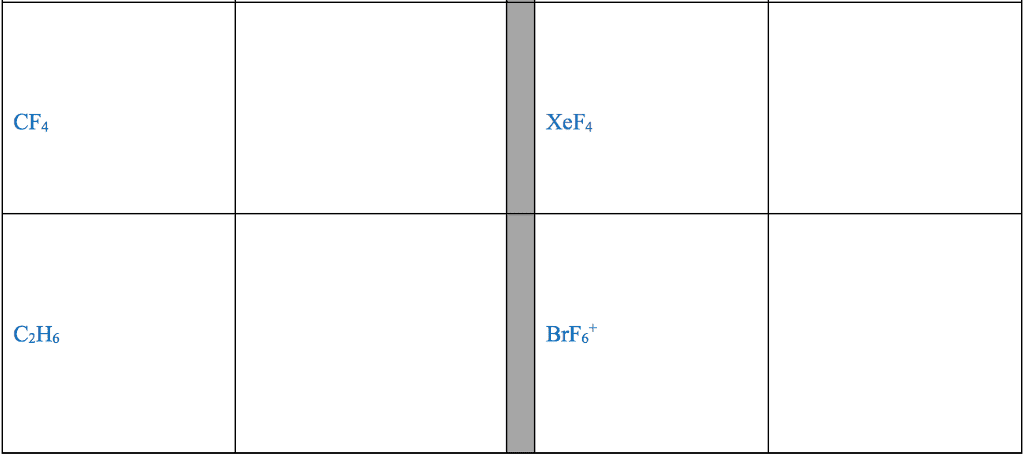
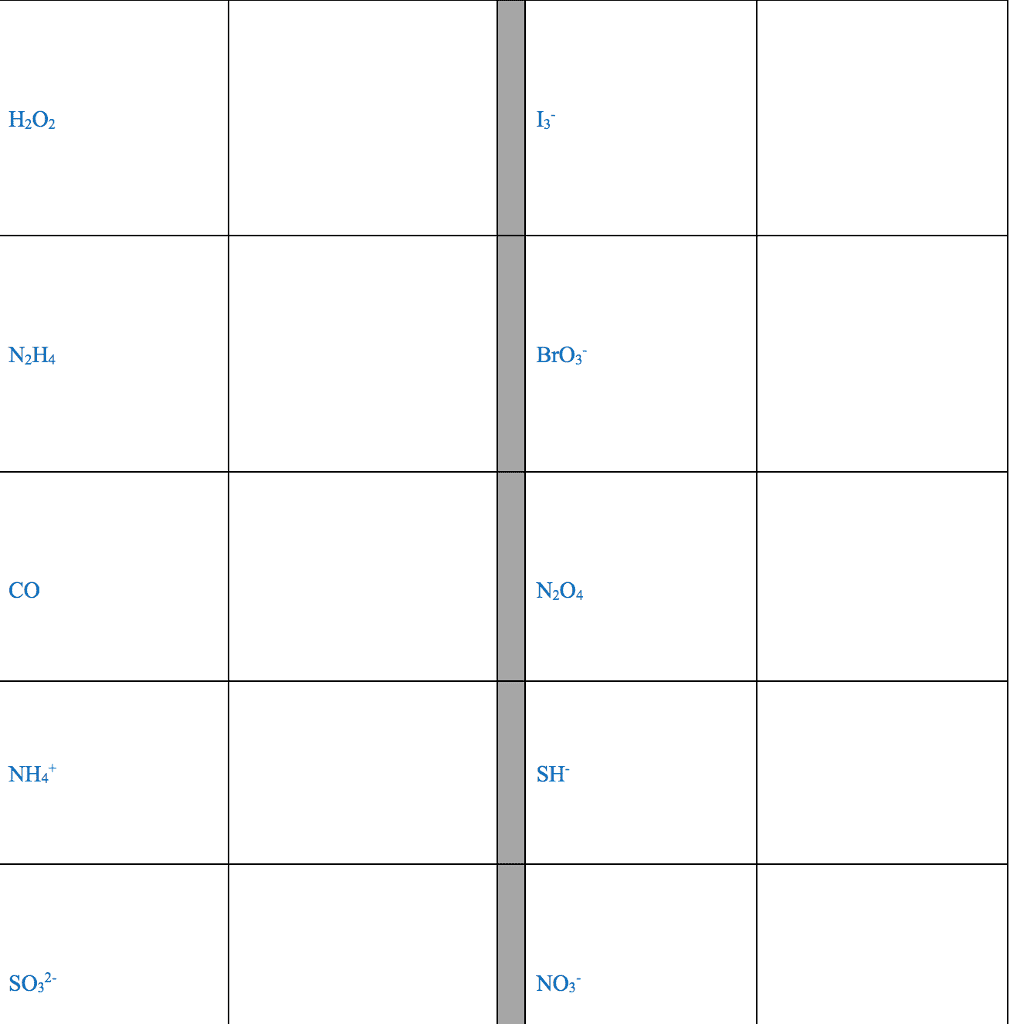
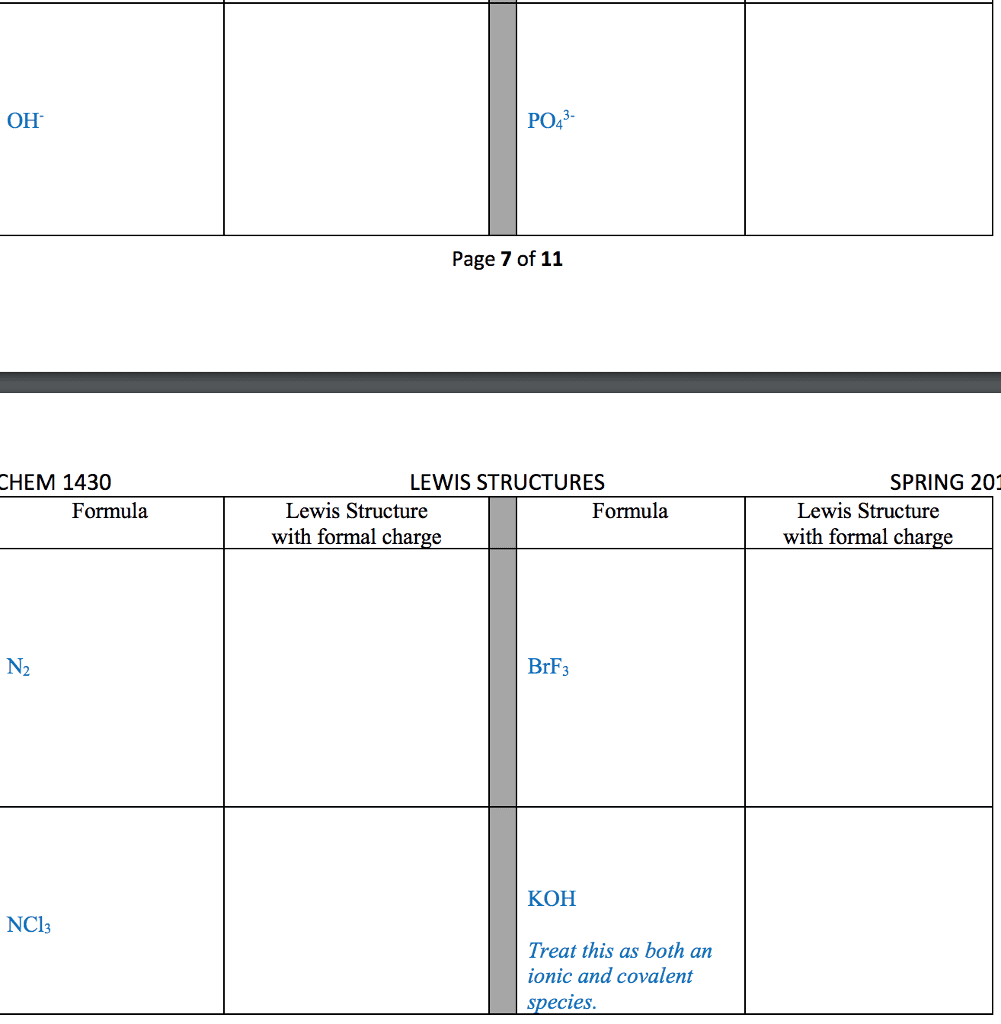
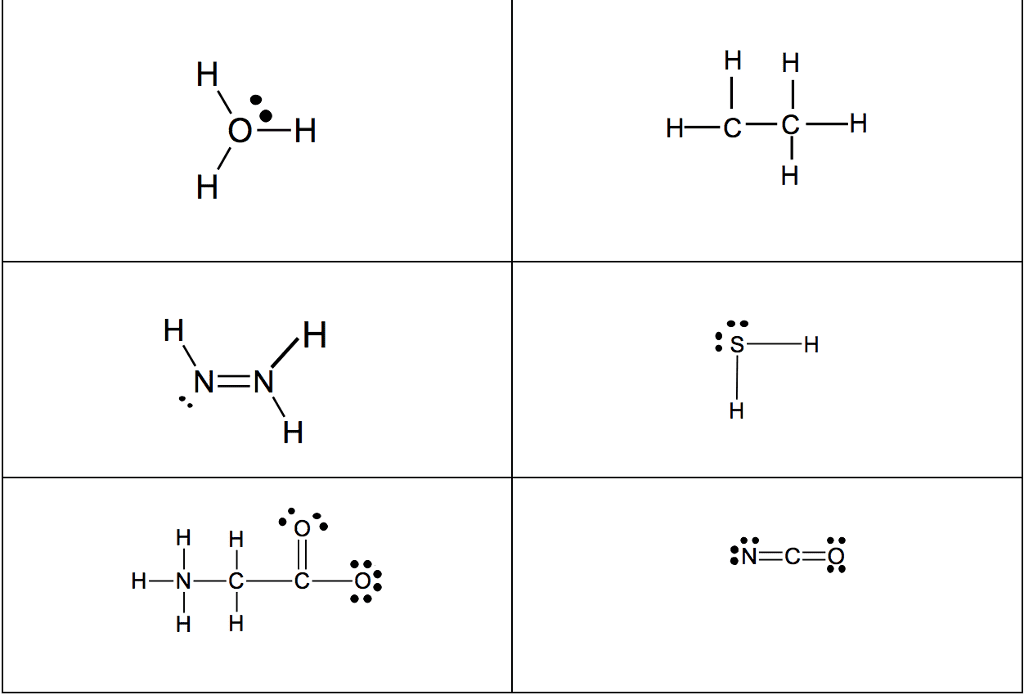
CHM 2045 Midterm: Exam 3 Notes
Document Summary
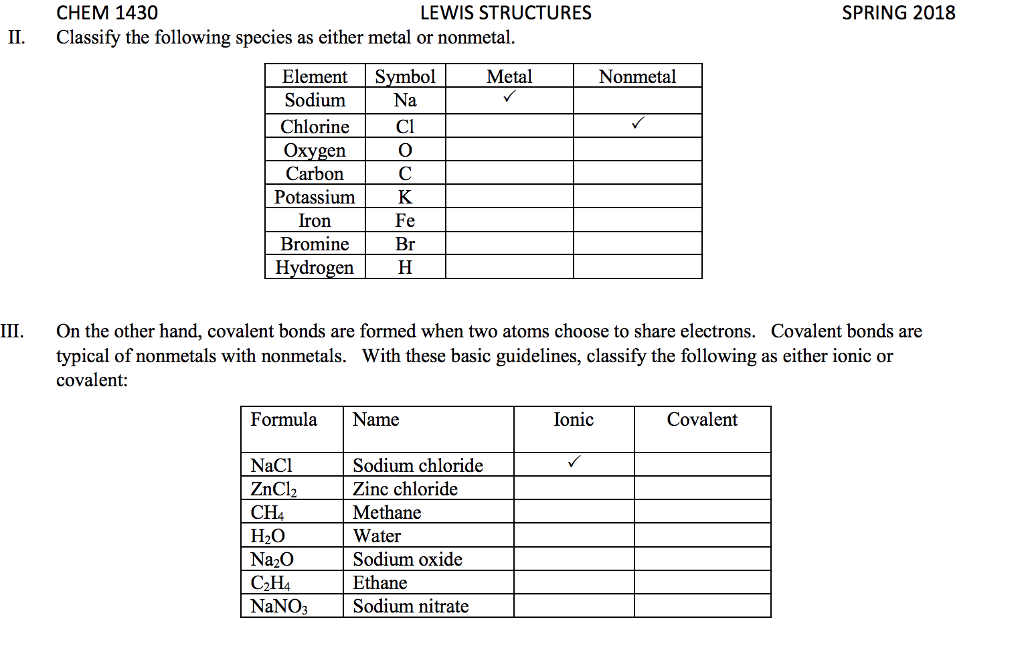
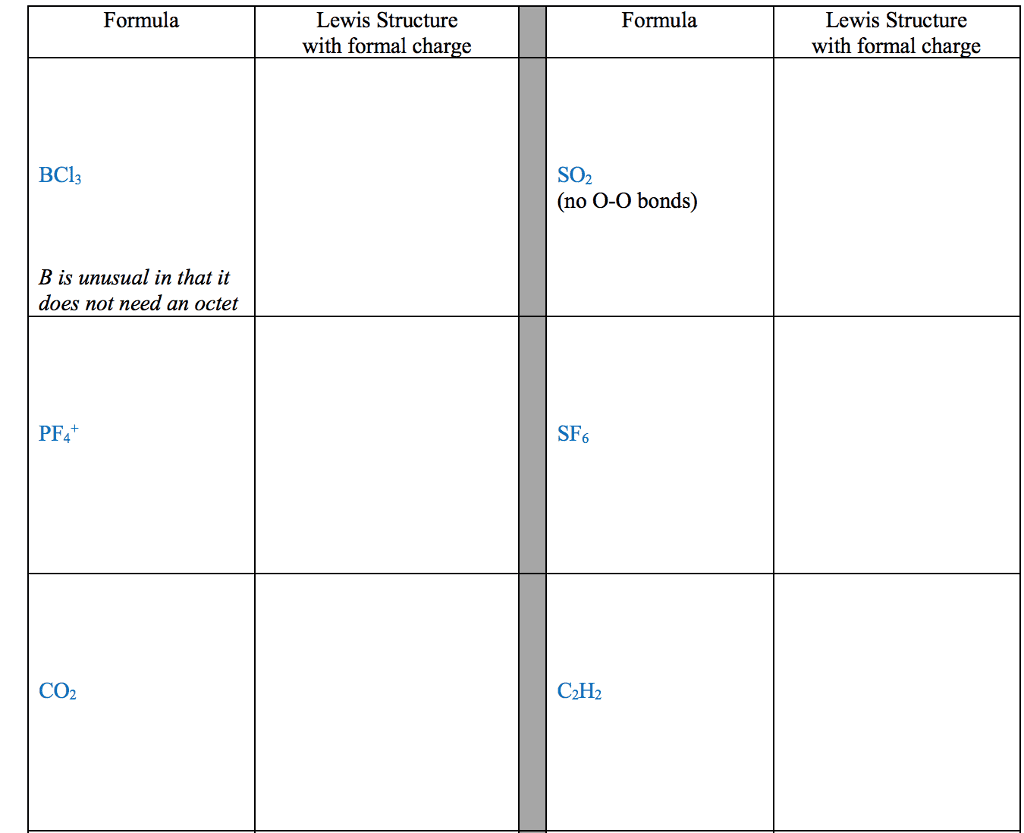
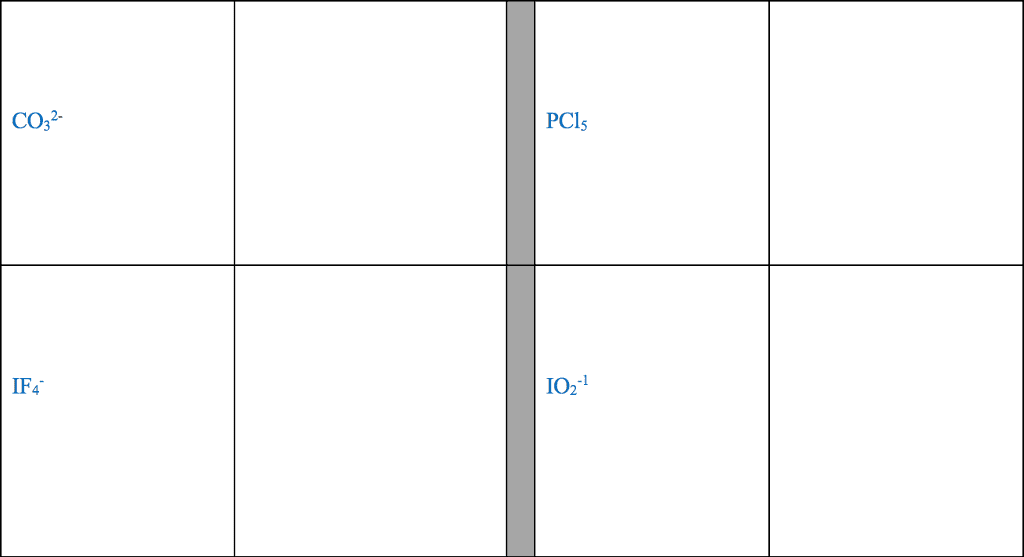
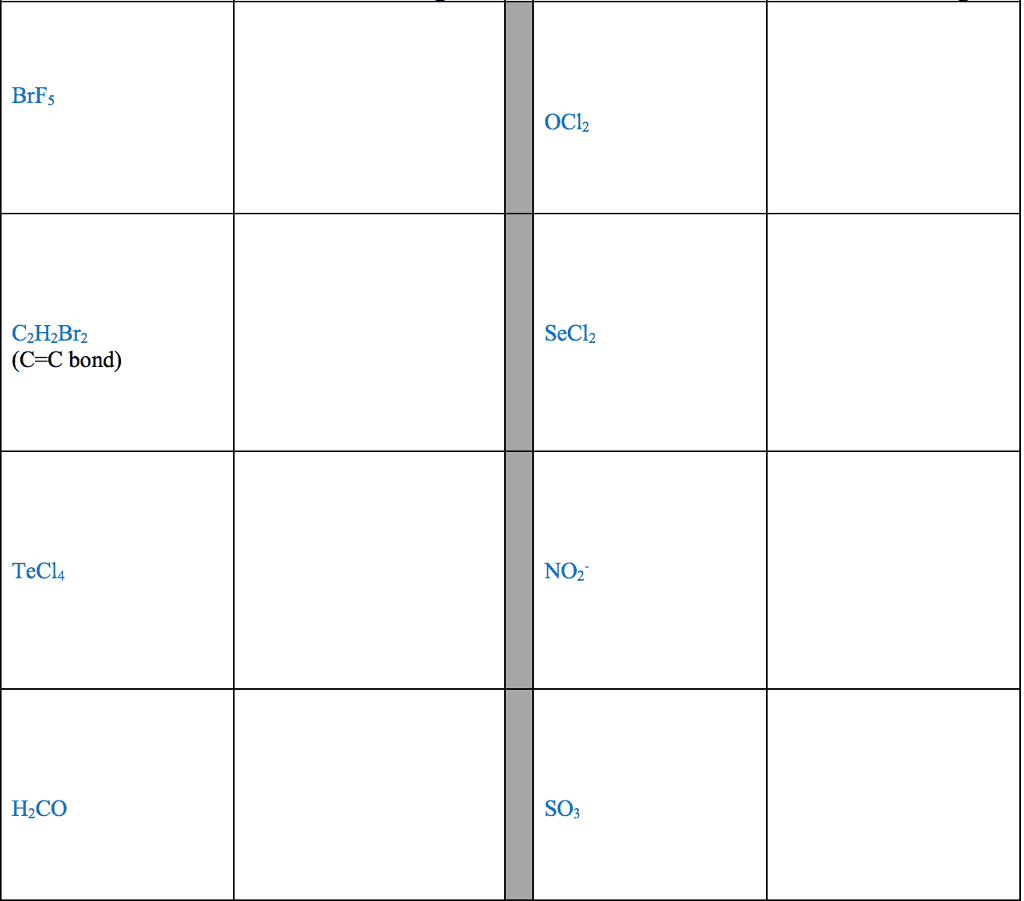
Review from chapter 9- very important to understand these concepts: The octet rule is the tendency of an atom to achieve a full valence shell by sharing electrons to achieve a noble gas con guration. The noble gas con guration is a stable state of an element when it has the same number of electrons as its nearest noble gas element. Lewis dot notation portrays the arrangement of valence electrons between two elements when the elements come together in a covalent bond or in an ionic bond. A covalent bond is the sharing of valence electrons between two or more nonmetals. An ionic bond involves transferring electrons between a metal and a nonmetal. For the representative elements, the valence electrons are the electrons in the outer shell (valence shell) of the atom. The number of valence electrons an atom has directly correlates to the group number in the 1a 8a groups.