CHEM 1A03 Chapter Notes - Chapter 5: Stoichiometry, Covalent Bond, Chemical Polarity

35
CHEM 1A03 Full Course Notes
Verified Note
35 documents
Document Summary
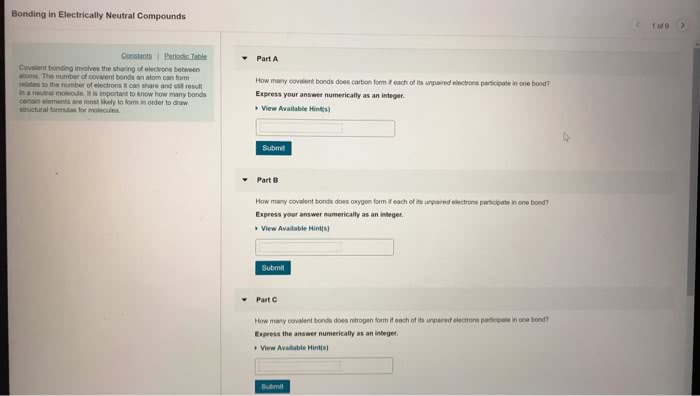
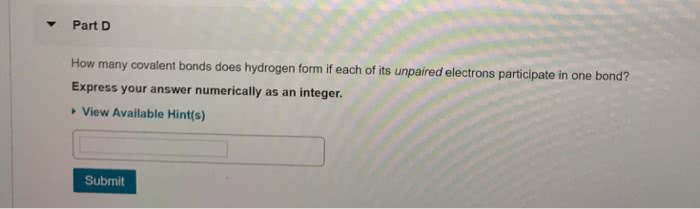
Distinguish ionic from covalent bonding and determine bond polarity. Draw resonance structures and calculate average bind order. Transfer of electrons from one atom to another. Lattice enthalpy: the energy released when we take a stoichiometric number of moles of ions in the gas phase and combine them into one mole of salt, in the solid state. If two electrons are being shared between the atoms, they can be represented by a line. Coordinate covalent bonds: one atom provides both of the electrons to form a bond (ex: ammonia reacts with a proton, h+. The result of this is the ammonium cation) Direction is indicated using a polar arrow and partial charges. Electrons are pulled towards the more electronegative atom. If the difference in electronegativity is very small or zero, there is a pure covalent bond. Percent ionic character increases with increasing difference in electronegativity.