CHMB16H3 Chapter Notes - Chapter 17: Chemiluminescence, Derivatization, Cuvette
Document Summary
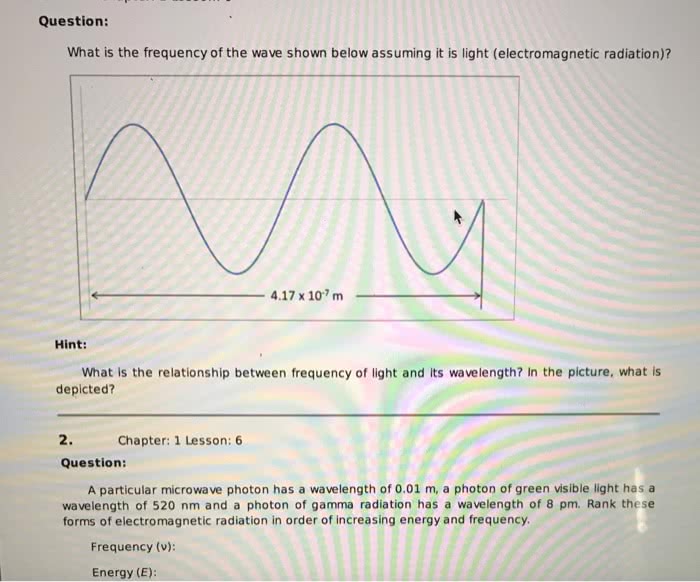
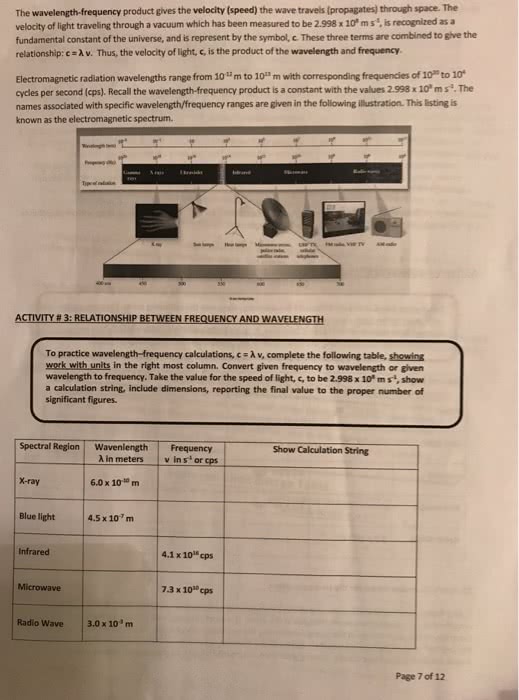
Spectrophotometry: any technique that uses light to measure chemical concentrations: colorimetry: a procedure based on absorption of visible light, wavelength ( ): the crest-to-crest distance between waves. For visible wavelengths in most substances, n > 1, so visible light travels more slowly through matter than through vacuum. Photons: particles of light with regard to energy: relation between energy and frequency: e = hv, where h is planck"s constant of. Irradiance/intensity/radiant (p): the energy per unit time per unit area in the light beam (w/m2): monochromator: a wavelength selector in which light passes through and it selects one wavelength. Transmittance (t): the fraction of original light that passes through the sample (t = P/po) and has a range of 0 to 1. Proteins are normally assayed in the ultraviolet region at 280 nm because aromatic groups present in virtually every protein have an absorbance maximum at 280 nm: reagent blank: contains all reagents but with analyte replaced by distilled water.