CHMA10H3 Chapter Notes - Chapter 7: Motal

36
CHMA10H3 Full Course Notes
Verified Note
36 documents
Document Summary
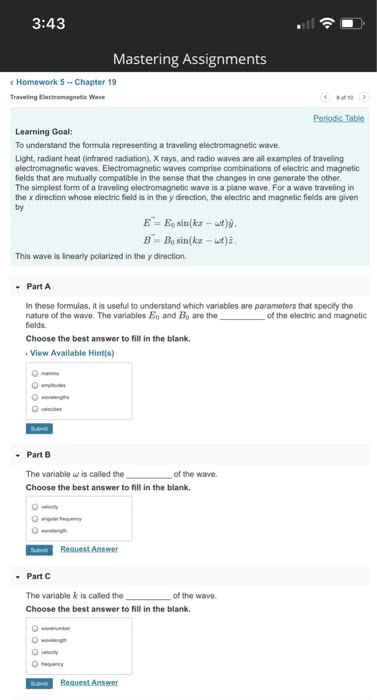
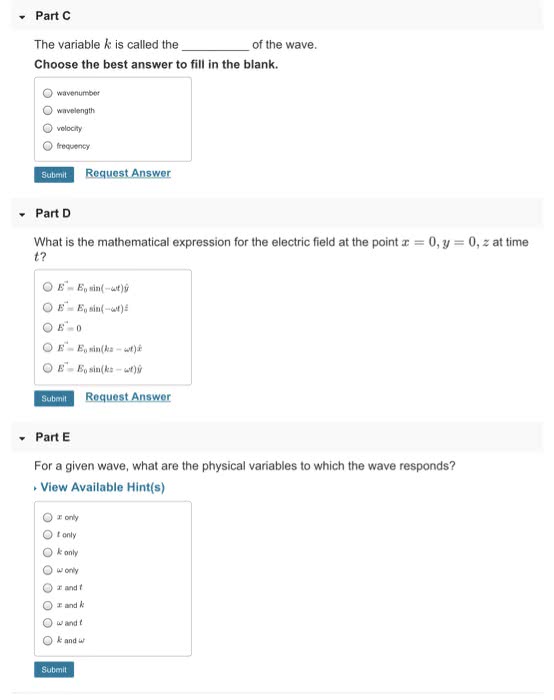
Chapter 7 the quantum- mechanical of the atom. 7. 1 quantum mechanics: the theory that explains the behaviour. Quantum-mechanical model: a model explaining how electron in atoms and how those electrons determine the chemical and physical properties in elements. Classical physics: at the beginning of the 20th century, matter was described as discrete particles, electromagnetic radiation were continuous waves, the two were thought to be separate entities. Light is electromagnetic radiation (a type of energy embodied by oscillating electric and magnetic fields) Electric and magnetic fields spread as waves through empty space or through a medium, a wve transmits energy, produced by accelerating electrically charged particles visible light (electrons) and radio wave (fluctuations of electric current) ): distance between tops of 2 successive crests/troughs, unit in. Amplitude: the maximum height of the wave. Frequency (v): number of crests/troughs passing through a given point/unit of time (s-1), unit: cycle/second. Velocity (c= speed of light) = 2. 9975 x 10^8 m. s-1.