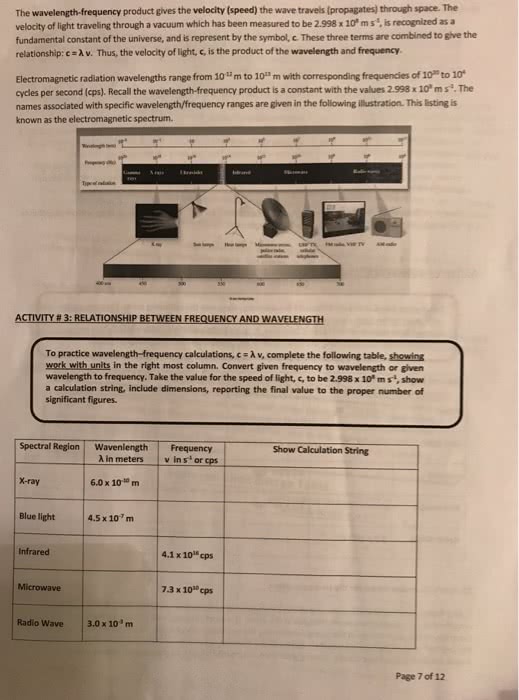
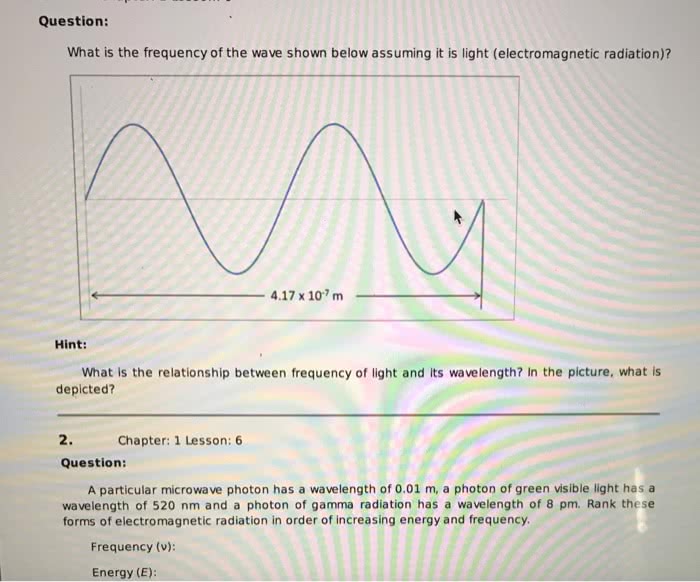
The wavelength-frequency product gives the velocity (speed) the wave travels (propagates) through space. The velocity of light traveling through a vacuum which has been measured to be 2.998 x 10" m s . is recognized as a fundamental constant of the universe, and is represent by the symbol, c. These three terms are combined to give the relationship:ãAv. Thus, the velocity of light, c, is the product of the wavelength and frequency. Electromagnetic radiation wavelengths range from 10 m to 101s m with corresponding frequencies of 10 to 10 cycles per second (cps). Recall the wavelength-frequency product is a constant with the values 2.998 x 10°m s. The names associated with specific wavelength/frequency ranges are given in the following illustration. This listing is known as the electromagnetic spectrum. ACTIVITY # 3: RELATIONSHIP BETWEEN FREQUENCY AND WAVELENGTH To practice wavelength-frequency calculations, c-λ v, complete the following table, showing work with units in the right most column. Convert given frequency to wavelength or given wavelength to frequency. Take the value for the speed of light, c, to be 2.998 x 10 ms ,show a calculation string, include dimensions, reporting the final value to the proper number of significant figures Spectral Region Wavenlength Frequency Show Calculation String λ in meters A in meters vin s or -is-or x-ray 6.0 x 10m Blue light 4.5 x 107m Infrared 4.1 x 10 cps Microwave 7.3 x10 cps Radio Wave 3.0x 10' m Page 7 of 12