CHM136H1 Chapter 1-4: Chapter 1 -4, 9

34
CHM136H1 Full Course Notes
Verified Note
34 documents
Document Summary
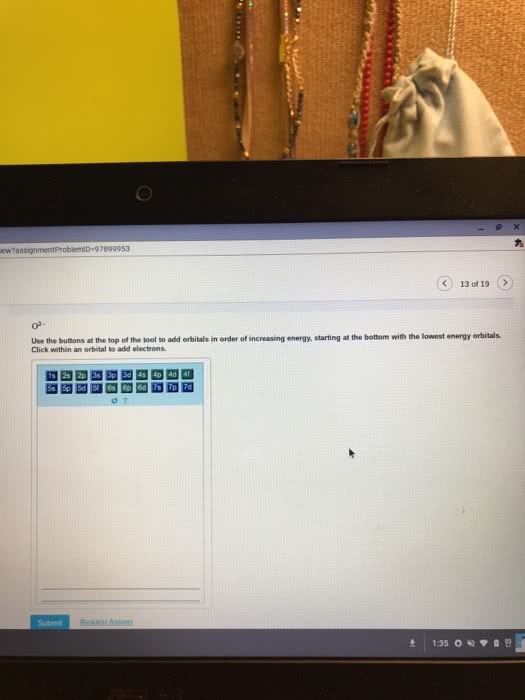
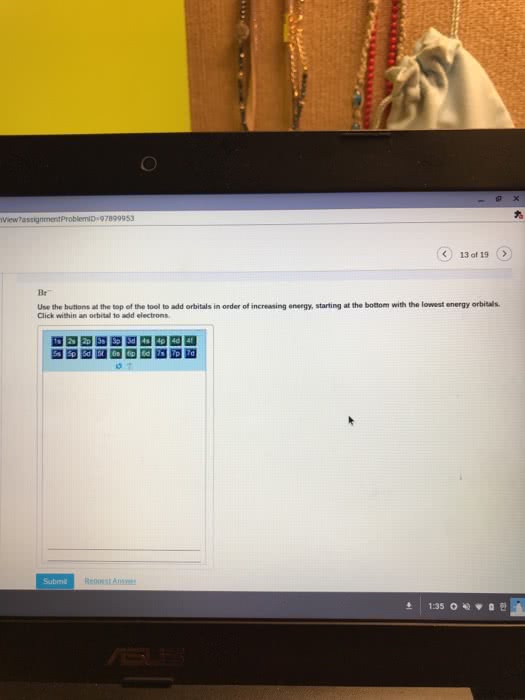
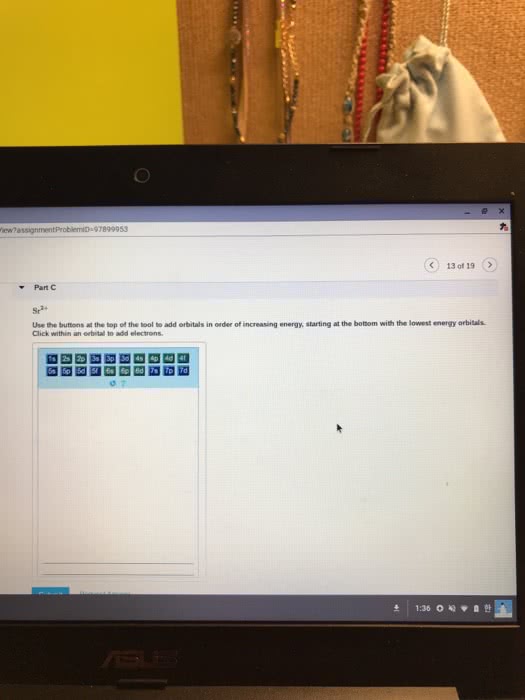
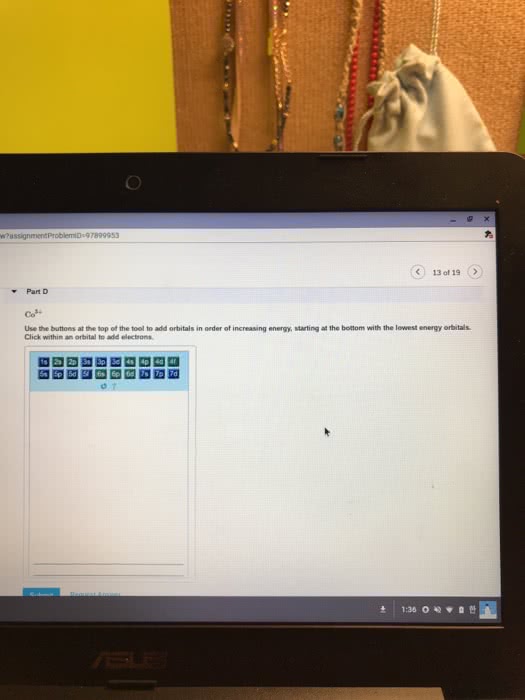
The addition of an electron is always placed on the lowest possible energy orbital. Pauli exclusion principle: no two electrons in a same atom have identical electron configuration. Electrons spins in opposite orientation, and . Hund"s rule: electron always half-fill all the orbitals in a energy level before fully fill the orbitals. Carbon: sp3, tetravalent (tetrahedral structre), 109. 5 sp2, t rivalent (trigonal planar), 1 p orbital, 120 sp, linear, 2 p orbitals, 180. Carbocation: sp2, no lone pair, valence electron: 3. Carboanion: sp3, 1 lone pair, valence electron: 5. Nitrogen: sp3, t rivalent, 1 lone pair, 109. 5 oxygen: sp3, divalent, 2 lone pair, 109. 5. Phosphorous: sp3, tetravalent * could have hybridization involving d orbitals, and also expand octect valence. **note: any heteroatom next to a sp2 hybridized carbon is also sp2 hybridized. Double bond: hybridized spn bond + 1 pi bond. Degree of unsaturation: (saturated # of hydrogen actual # of hydrogen)/2.