Textbook Guide Chemistry: Macrocycle, Mitosis, Reagent

34
CHM136H1 Full Course Notes
Verified Note
34 documents
Document Summary
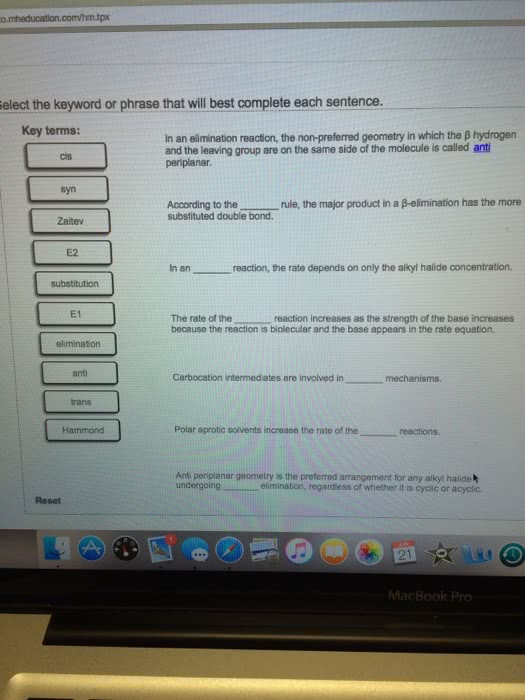
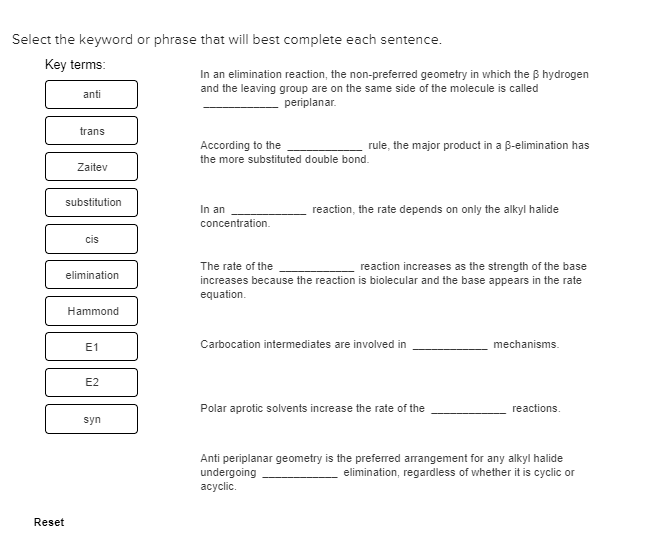
These are the types of reactions in which conversion of reactant into product takes place in a single step. The conversion of ethene in the presence of molecular bromine to form vicinal dibromide or 1, 2-dibromoethane is an example of one step synthesis reaction. The medium of the reaction is a solvent which is ccl4. The change of position of a base can be done by adding alcoholic koh to an alkyl halide which is referred to as an elimination reaction as it attacks the beta hydrogen of the alkyl halide. In the second step the halogen acid such as hcl is added to the alkene formed in the eliminations step which results in an alkyl halide in which the position of base has changed. In the above reaction, two products can be formed but the major product is the more substituted alkene because it is the one whose hydrogen has been attacked.