Chemistry 1027A/B Chapter : Chemistry 1100A Notes .docx
Document Summary
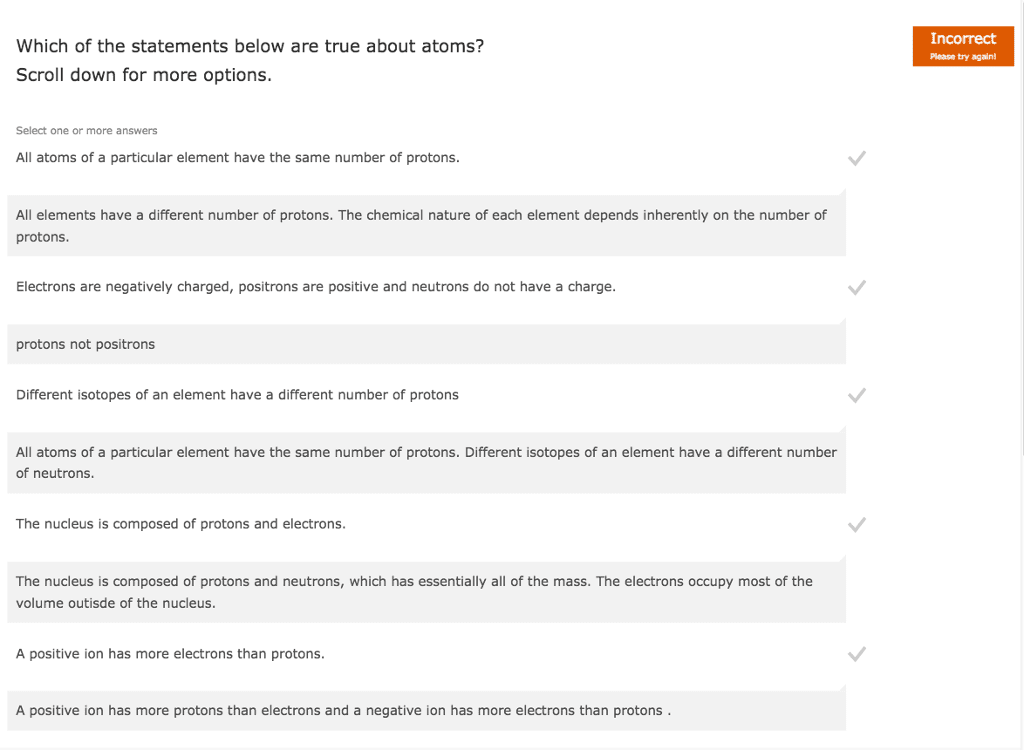
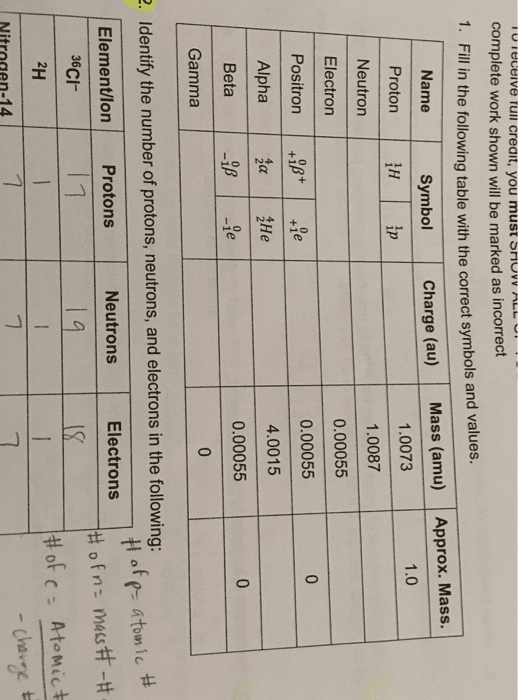
Atoms building blocks of all elements which contain 3 main components: proton, The mass is equal to one atomic mass unit. Makes up most of the mass of the atom. Make up most of the mass of the atom. Mass number = # protons + # neutrons. Atomic number the number of protons in an atom. Mass number total number of protons and neutrons in an atom. Isotopes atoms with the same number of protons but different numbers of neutrons. Isotopic abundance percent by number of each of the isotops of an element. The atomic mass on the periodic table of an element tells us the average value of the masses of all isotopes of that element. Ex: silicon has 3 isotopes (si-28, si-29, si-30) Si-28 is most abundant in the world so the average atomic mass (aka amu or. U ) is closer to 28 u rather than 29 or 30 u.