Chemistry 1301A/B Chapter Notes - Chapter General Review: Exclusive Or, Molecular Mass, Eaves
Document Summary
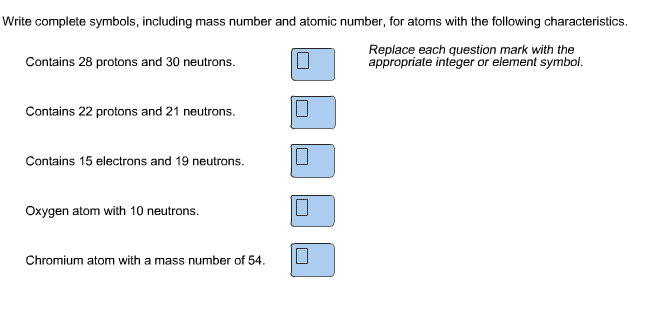
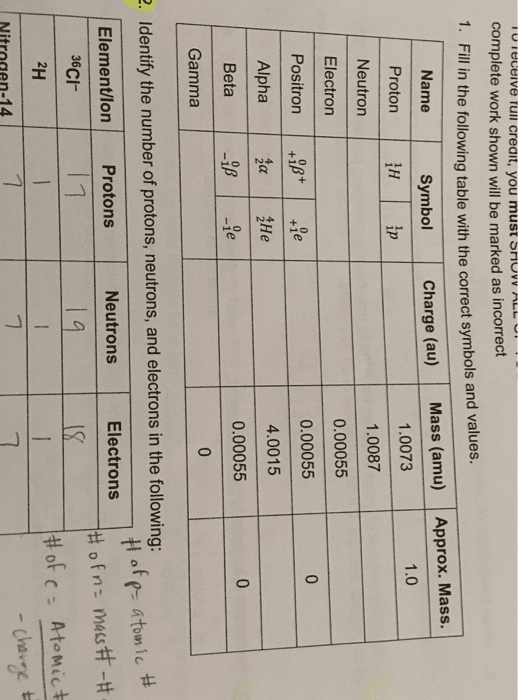
The building blocks of matter that consist of protons (+), electrons (-), and neutrons (neutral) Protons and neutrons are found in the atom"s nucleus, while electrons occupy the space around the nucleus. They also have almost the same mass and make up most of the mass of the atom. Mass of electrons = 1/1836th of mass of a proton or neutron. Atomic number (z) the # of protons in a given element. All the atoms of the same element have the same number of protons (i. e. the z of carbon is 6 and every carbon atom has 6 protons) Mass number (a) = (# of protons in atom) + (# of neutrons in atom) Always has an integer value (1, 2, 3,) These notations are often used : or -, where represents a chemical symbol. Atomic mass the physical mass of an atom expressed in grams (g), kilograms (kg, or other units.