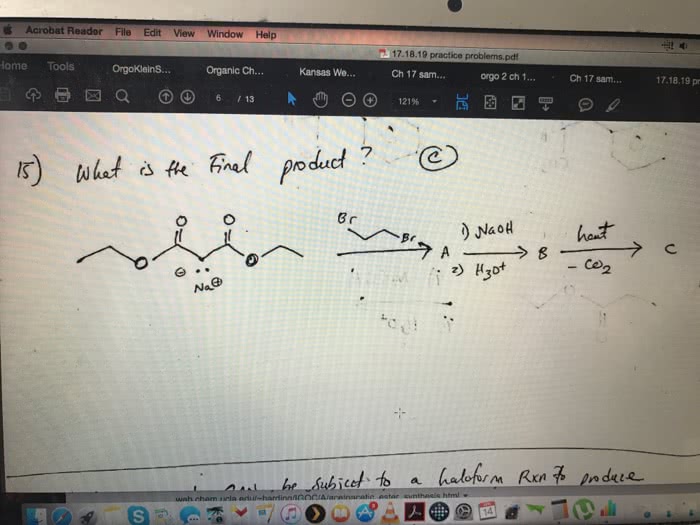
Please answer the 2nd to last question. I think the answer should have the potential energy diagrams. one sentence is not a sufficient answer. needs to be detailed.

Consider the following reactions of ethylbenzene (PHCHzCHHwith chlorine or iodine: (Ph--Colls) + "CI PhCH-CH + HCI 10 + I PhCHCHL + H-1 10 PhCH-CH-H PhCH-CH-H + CI PhCH-CH3 + HCI 2 + I PhCH-CH3 + HI 2° PhCH-CHDH'Olion 356 HHC1 431 HH 297 Using the bond dissociation energies given below, calculate the AH for each of the four pathways. Oph - CH-CH, -At CI ph-car H - C1 A -B c410-431 Car Ho-H - Hr PhCHCHH 410 9 ph -cr -Crg Tel. +phy CH -CH, + HCI 2. 15 Timo1 DH a sh D Ha-35 , S SD - 31 - C ® ph-CHI-CH, -H rI⢠pm -cha-c, H-1 Hons A Me -- on a -14 no-aa1=23 PTI mo) © crcH3 + circhs H-I ph - - 3 * h - On nono One 35 - 20 - 59 as I mol Which reaction (chlorination or iodination) is more thermodynamically favorable? Why? Favorable since chlorination is not thermodynami coly it is exonermit because it is negative. Which intermediate free radical (1° PhCHCH or 2°/benzylic PhCHCH) is more stable? Show why. The intermediate ph- cR- cts is more stable Since 1 ci-Cng is stabilized by resonance in pney ring which reaction (chlorination or iodination) will be more selective for substitution at the secondary (benzylic, Ph-CHW) carbon than at the primary carbon (-CH)? Using reaction vs potential energy profiles, explain in terms of the Hammond-Lefler postulate. (Use the backside of this page for your answer) What would be the structures) of the final products of the chlorination and iodination reactions? - Chiorination - ph - CH - CH, + PN-CHе - CH Lodington -ph-CH-CAg PM- CH4 CH, I