Part, A ,and B
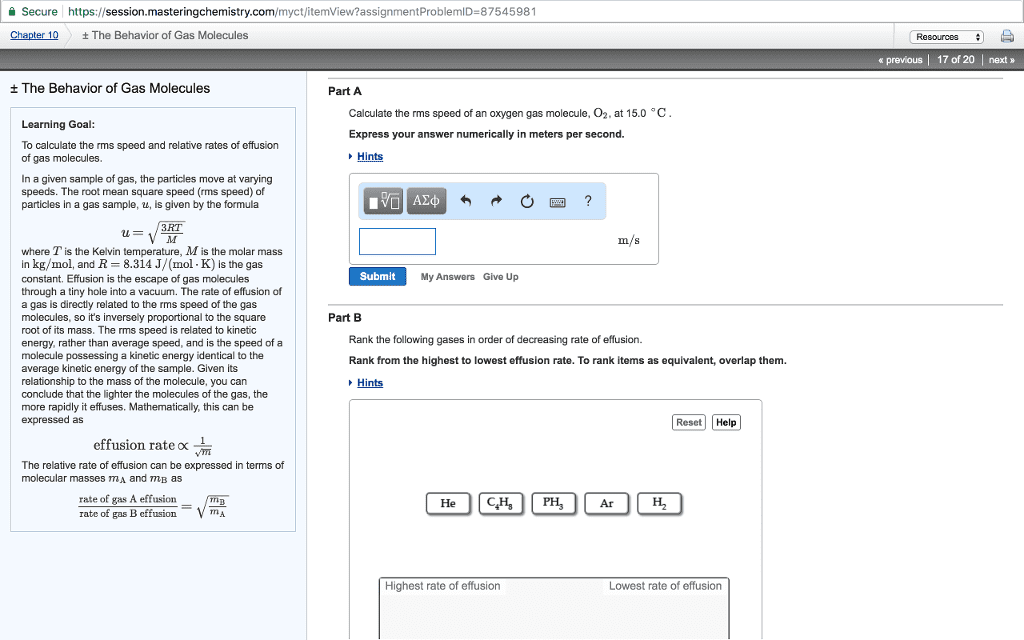
a secure https://session.masteringchemistrycom/myctitemView?assignmentProblemID=87545981 Chapter 10 + The Behavior of Gas Molecules Resources a previous 17 of 20 next > + The Behavior of Gas Molecules Part A Calculate the rms speed of an oxygen gas molecule , O2, at 15.0 °C Learning Goal: Express your answer numerically in meters per second. To calculate the rms speed and relative rates of effusion of gas molecules. Hints In a given sample of gas, the particles move at varying speeds. The root mean square speed (ems speed) of particles in a gas sample, u, is given by the formula DVD AP to O o ? 3 RT m/s Submit My Answers Give Up Part B where T is the Kelvin temperature, M is the molar mass in kg/mol, and R = 8.3i4 J/(mol -K) is the gas constant. Efusion is the escape of gas molecules through a tiny hole into a vacuum. The rate of effusion of a gas is directly related to the rms speed of the gas molecules, so it's inversely proportional to the square root of its mass. The ms speed is related to kinetic energy, rather than average speed, and is the speed of a molecule possessing a kinetic energy identical to the average kinetic energy of the sample. Given its relationship to the mass of the molecule, you can conclude that the lighter the molecules of the gas, the more rapidly it effuses. Mathematically, this can be expressed as Rank the following gases in order of decreasing rate of effusion. Rank from the highest to lowest effusio on rate. To rank items as equivalent, overlap them . Hints ResetHelp effusion rate a m The relative rate of effusion can be expressed in terms of molecular masses mA and mB as rate of gas A effusion rate of gas B effusion - He CHPH,AH ma Highest rate of effusion Lowest rate of effusion