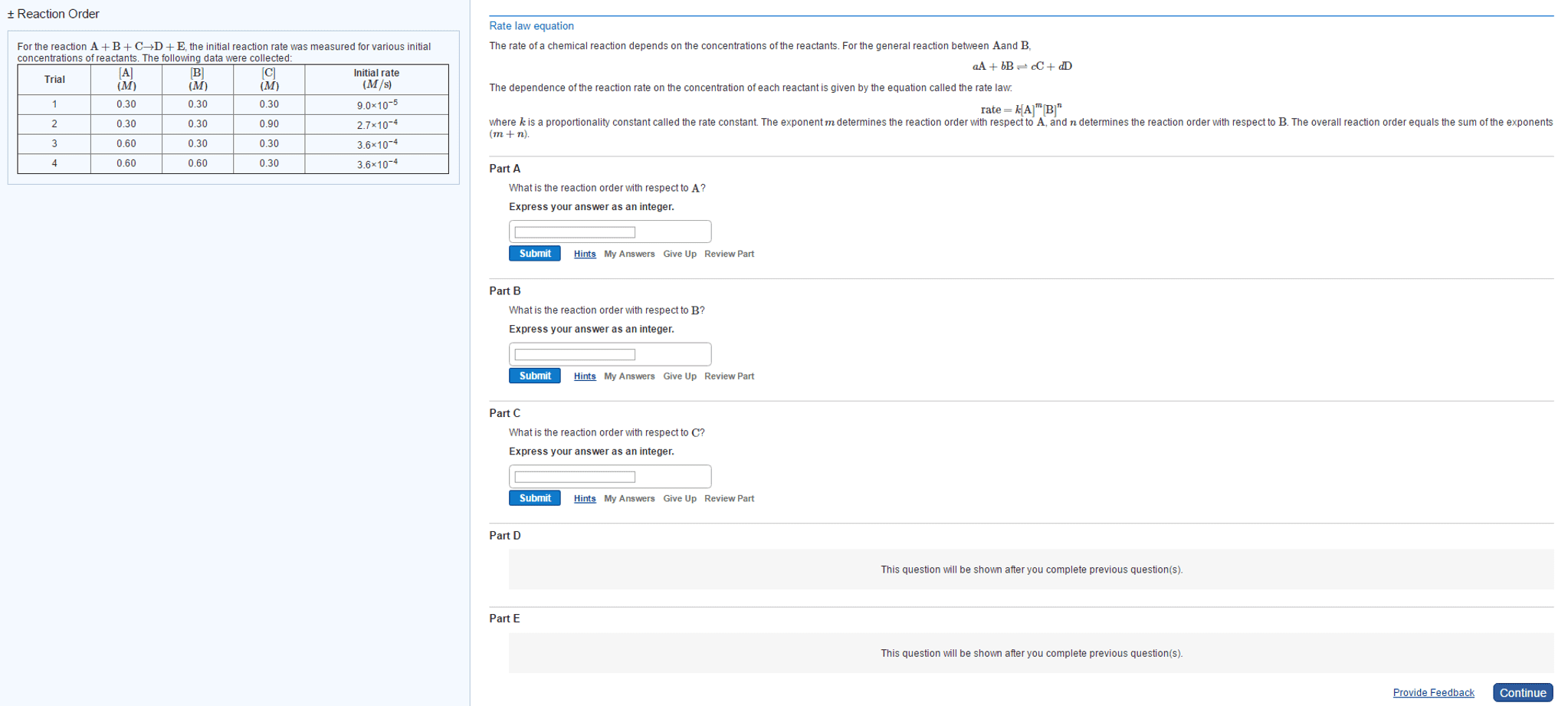
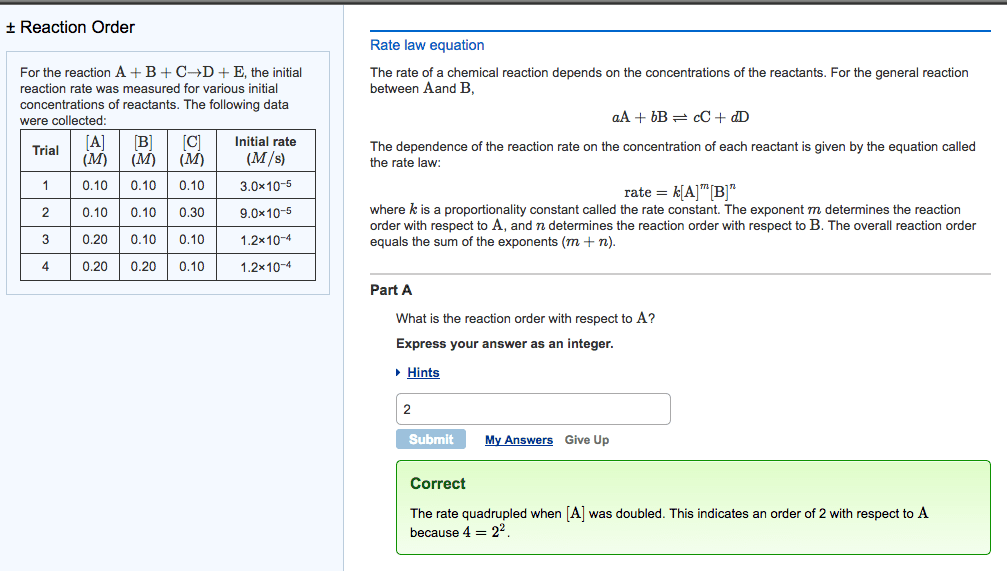
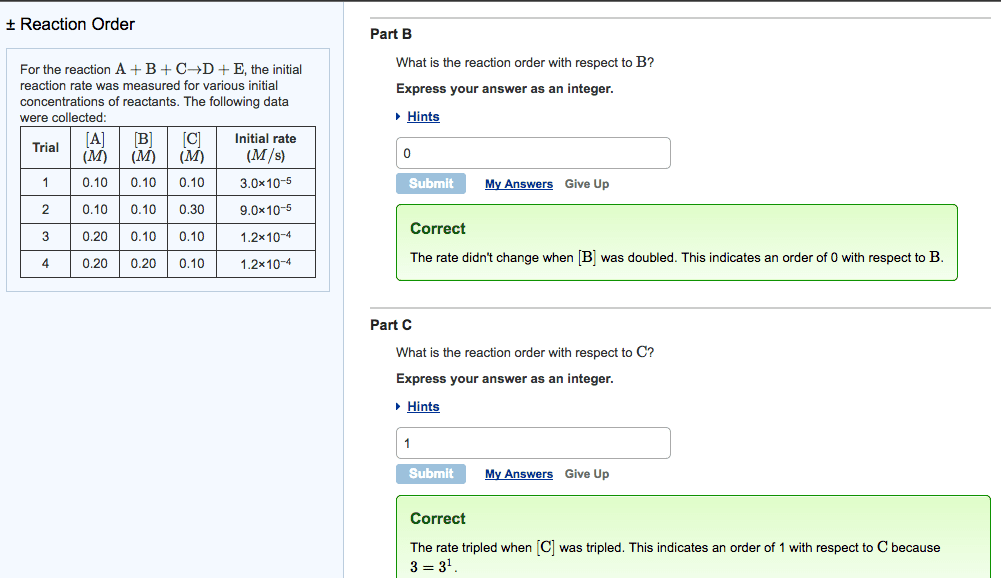
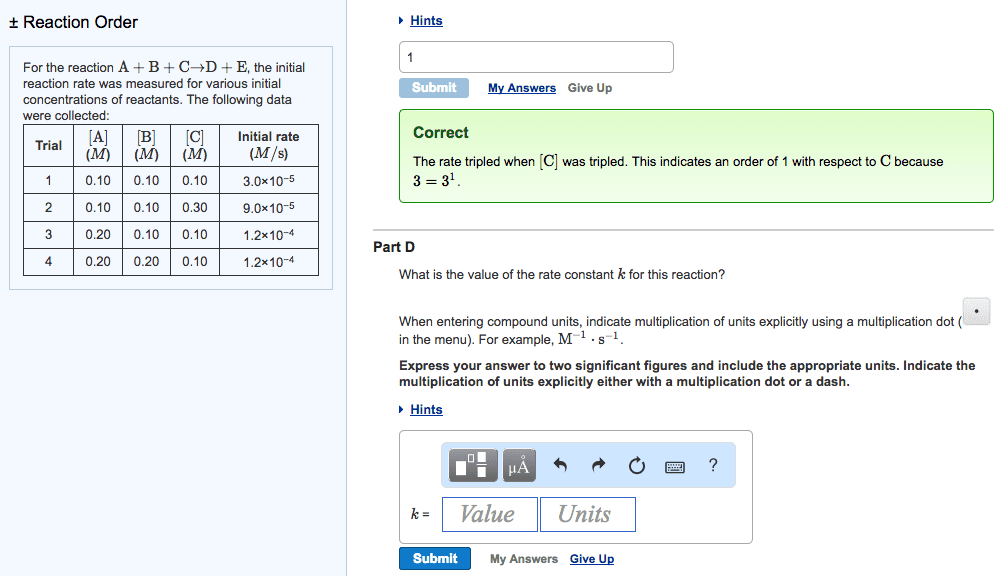
CH-1020 Chapter Notes - Chapter 13: Thermodynamics, Reaction Rate Constant, Stoichiometry
Document Summary
Reactions occur when reactants collide in the correct orientation, with enough energy. The rate (speed) of a given chemical reaction depends on concentration of reactants. Reaction kinetics studies are done to gain information about how the reaction proceeds (number of steps, order of steps, relative rates of each step (cid:863)reactio(cid:374) (cid:373)echa(cid:374)is(cid:373). (cid:863)) Given rate law, determine overall order and order wrt to each reactant. Interpret and draw reaction profiles (reaction coordinate diagrams) Add this to your homework: ch 13 terms to look up and know. 0, 1st, and 2nd order reactions: rate laws, graphs, integrated forms. A chemical equations shows what reactants yield what products and give mole ratios. Thermodynamics predicts whether a reaction will proceed as written. Kinetics describes describes the reaction rate and reaction mechanism, encompasses the how and how fast concentration. Numerator is change in sufficient energy in the correct orientation. Higher temperatures often accelerate the reaction (make it faster).