CHEM 1220 Chapter Notes - Chapter 15: Endothermic Process, Equilibrium Constant, Haber Process
Document Summary
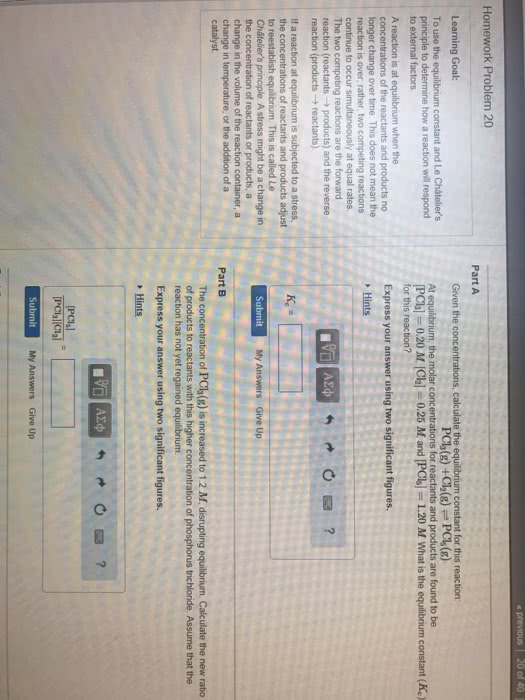
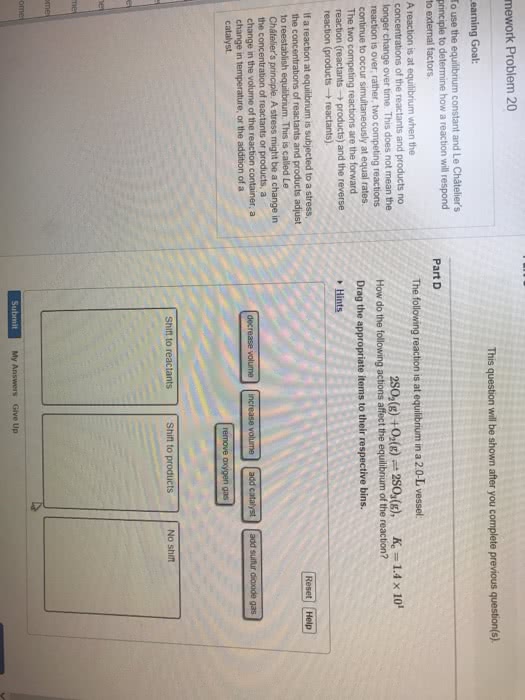
Chemical equilibrium: when opposing reactions proceed at equal rates. Equilibrium state: a mixture of reactants and products whose concentrations no longer change with time. Equilibrium is dynamic, reaction doesn"t stop, just settles into a given ratio. Equilibrium can be analyzed using kinetics and reaction rates. Equilibrium can be found by isolating the reaction rates from the forward and reverse reactions (k f/kr) For the equilibrium to be maintained, no reactants or products can escape from the system. Haber process: where n2 and h2 react at high pressure and temperature with the presence of a catalyst to form ammonia. Important with compost in the food industry; when in closed system, not all the reactants are used, maintain an equilibrium. Equilibrium can be reached from either direction of the chemical equation. Equilibrium constant expression: for equation aa + bb cc + dd. Equilibrium constant expression depends only on stoichiometry of the equation, not mechanism.