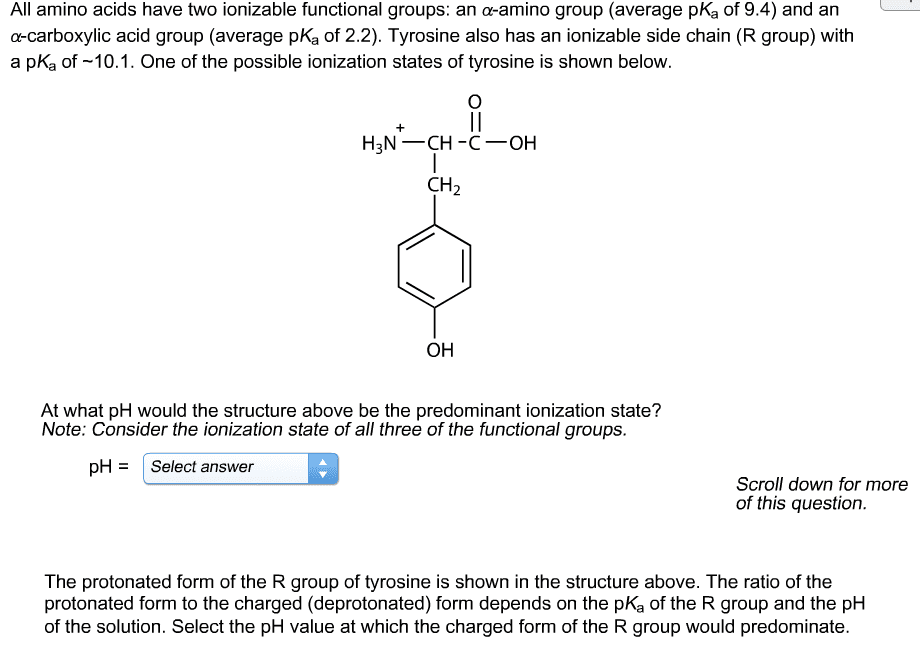
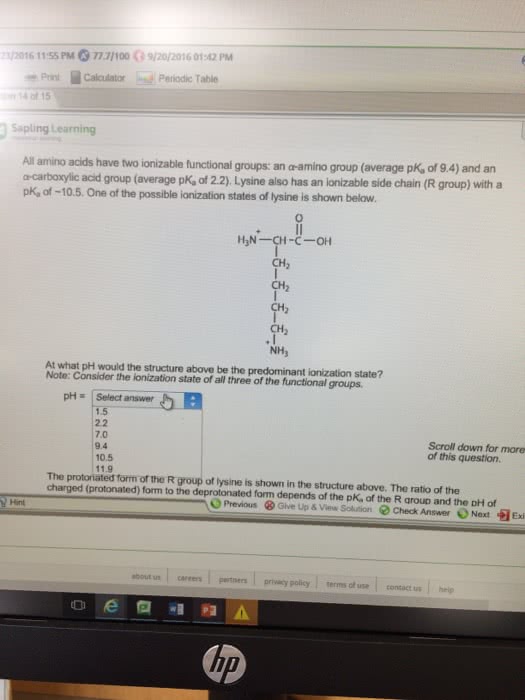
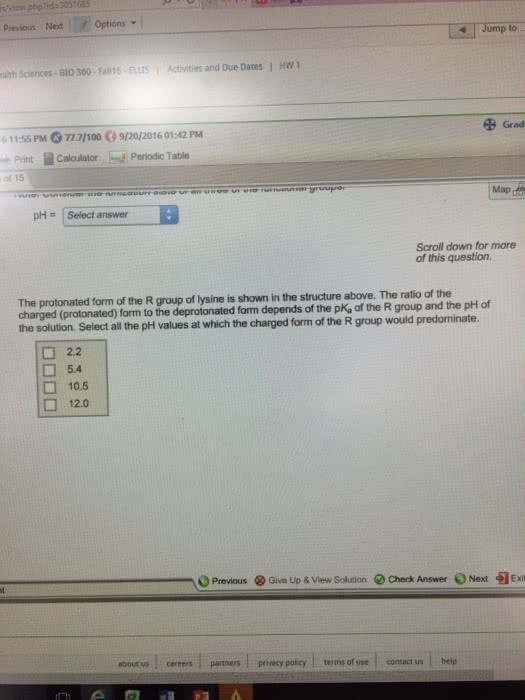
/2016 11:55 PM O 77.7/100ã9/20/2016 01:42 PM Print Calculator Periodic Table on 14 of 15 Sapling Learning All amino acids have two ionizable functional groups: an a-amino group (average p a-carboxylic acid group (average pK, of 2.2) Lysine also has an ionizable side chain (R group) with a pK, of -10.5. One of the possible ionization states of lysine is shown below. an a-amino group (average pK, of 9.4) and an K of 9.4) and an CH CH2 NH, At what pH would the structure above be the predominant ionization state? Note: Consider the ionization state of all three of the functional groups. pHSelect answer pH = | Select answer 1.5 2.2 7.0 9.4 10.5 11.9 Scroll down for more of this question. The protoiated form of the R group of lysine is shown in the structure above. The ratio of the charged (protonated) form to the deprotonated form depends of the pK, of the R group and the DH of Hint Previous ⧠Give Up & View Sohdon Check Answer Next Exi about us careerspertners privacy policy terms of use contact ushelp up