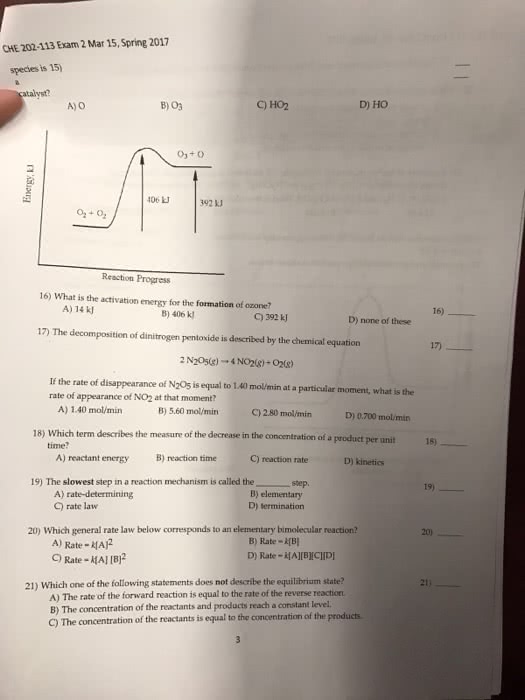
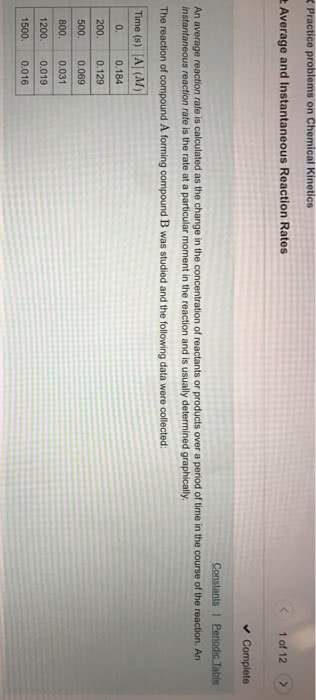
CHM 2046 Chapter Notes - Chapter 13: Spectrophotometry, Polarimetry, Third Order
Document Summary
Introduction: the speed of a chemical reaction is called its reaction rate. 1/(coefficient: rate = - [h2]/ t = - [i]/ t = (1/2) [hi]/ t. Average rate: the average rate is the change in measured concentrations in any particular time period, linear approximation of a curve, the larger the time interval, the more the average rate deviates from the instantaneous rate. Ions react faster than molecules: no bonds need to be broken, so less energy is needed. Increases the frequency of reactant molecule contact: concentration of gasses depends on the partial pressure, higher pressure = higher concentration, concentrations of solutions depend on the solute-to-solution ratio (molarity) We can then solve for k by plugging in one row of values and solving for k: rate = k[a]n[b]m, 0. 0021 = k[. 10]2[. 10]0, k = 0. 21 m-1s-1. Arrhenius equation: exponential factor: the exponential factor in the arrhenius equation is a number between 0-1. Increasing the temperature- increase the average kinetic energy, so.