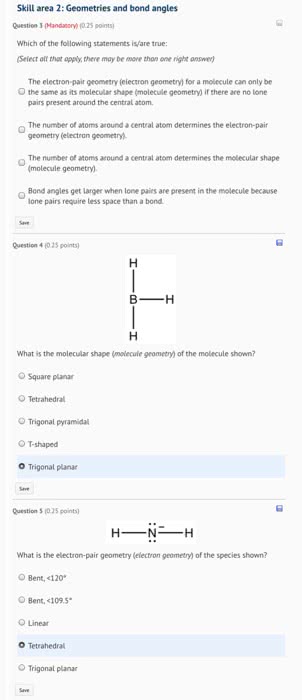
Practice Quiz CH 5 1. Which electron-pair geometry has the lowest electron-electron repulsive forces? a. linear b. trigonal planar c. tetrahedral d. trigonal bipyramidal e. octahedral 2. What is the steric number of the central phosphorus atom in PE,2 a. b. 3 d. 5 e. 6 3. Determine the molecular geometry of the carbonate ion, co? a. bent b. trigonal planar c. trigonal bipyramidal d. tetrahedral e. trigonal pyramidal 4. The electron group geometry of AsHs is , and its molecular geometry is d. tetrahedral; trigonal pyramidal. e. trigonal pyramidal; trigonal pyramidal. a. trigonal planar, trigonal planar. b. tetrahedral; trigonal planar. c. tetrahedral; tetrahedral. 5. Which compound below has the same molecular shape as CCl,H? a. SF b. KrF c. SiF e. CaH2 6. Which compound below has a square pyramidal molecular geometry? a. XeF b. IFs d. PFs e. SF 7. Which of these molecules have a dipole moment? CI CI 0 Cl Cl CI CI a. PCls and SiCl b. POCls, SO Cla, and SO c. POCls and SO2Cl d. PCls, POCIs, SOCI2, SOs, and SiCl e. PCIs, SF6, SOs, and SiCl