CHEM 1062 Chapter Notes - Chapter 19.1-3: Buffer Solution, Conjugate Acid, Titration Curve
141 views3 pages
Document Summary
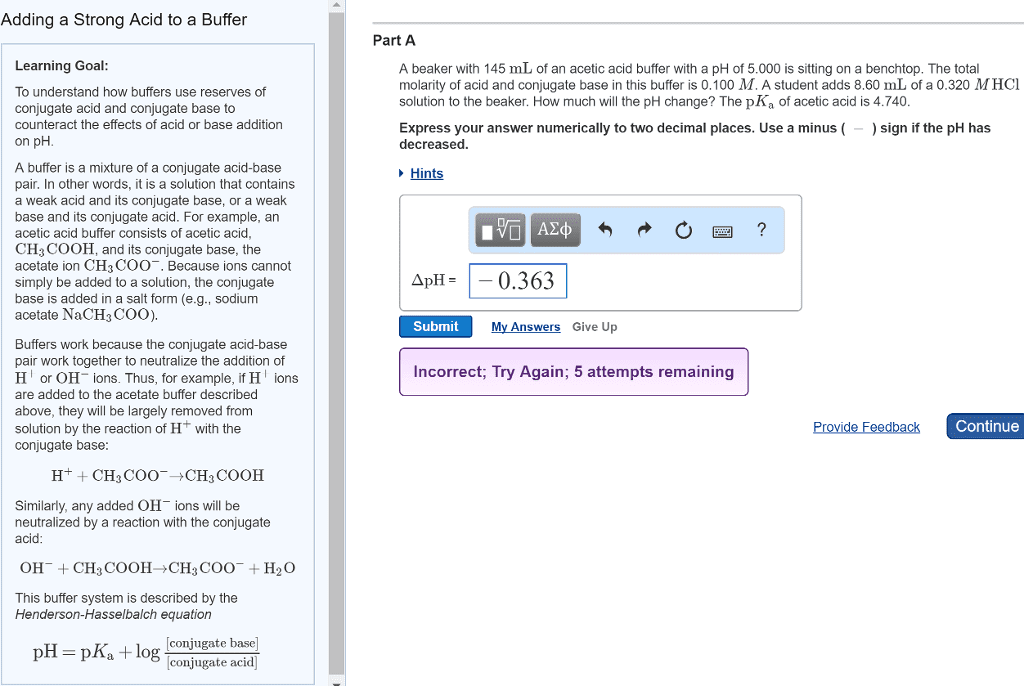
Buffer lessens the impact of an external force. An acid base buffer is a solution that lessens the impact of ph from the addition of an acid or base. To withstand the additions of h3o+ or oh-, a buffer must have an acidic component to react with the added oh- and a basic component to react with the added h3o+ The components are a conjugate acid base pair (weak acid + conjugate base or weak base + conjugate acid) Common ion effect occurs when a given ion is added to an equilibrium mixture that already contains that ion & the position of the eq shifts away from forming it. Common ion a- suppresses the dissociation of ha which makes the solution less acidic. Buffers work because large amounts of the acidic (ha) and basic (a-) components consume small amounts of added oh- and h3o+ respectively. If ratio [ha]/[a-] goes up, [h3o+] goes up.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232