CHEM 204 Lecture Notes - Benzaldehyde, Aldehyde, Ethyl Cinnamate
Document Summary
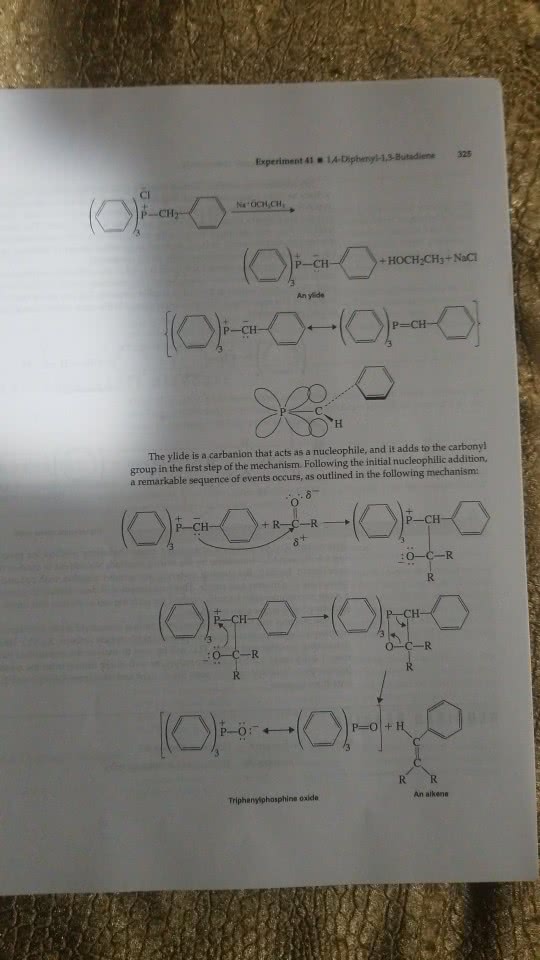
Experiment 5 (part 2): wittig reaction: objective: 1 mark. To perform the wittig reaction and synthesize ethyl cinnamate from ylid and benzaldehyde in the presence of the hexane solvent. Spectroscopy to verify if it is that product that is formed: introduction: 1 mark. Brief description of the concept/reaction studied wittig reaction. The wittig reaction involves the reacting ylide with an aldehyde or ketone, which is a carbonyl group, to form an olefin/alkene. The phosphonium ylid can be made through an sn2 reaction between the triphenyl phosphine and alkyl halide. When a stabilized ylid is used only alkenes are formed, while if an un-stabilized ylid is used alkenes and triphenyl phosphine oxide is formed. The wittig reaction is known to be a useful reaction since it produces a double in a specific position with no uncertainty (hunt, 2011): reaction equation: 1 mark.