BIOCHEM 2EE3 Lecture Notes - Thermodynamics, Enthalpy, Exergonic Process
Document Summary
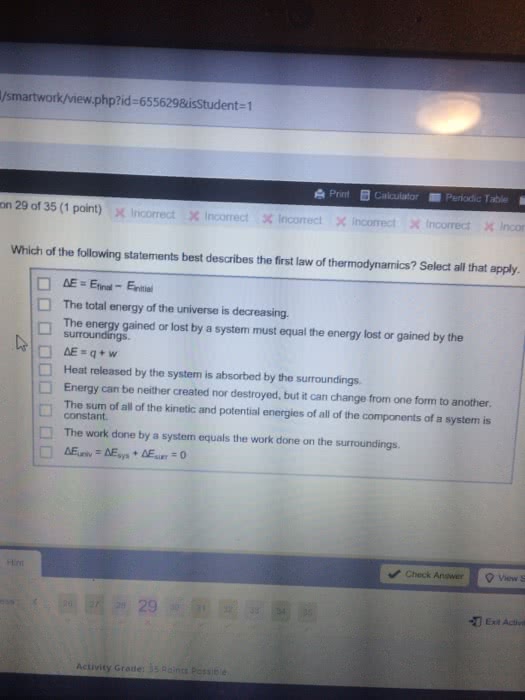
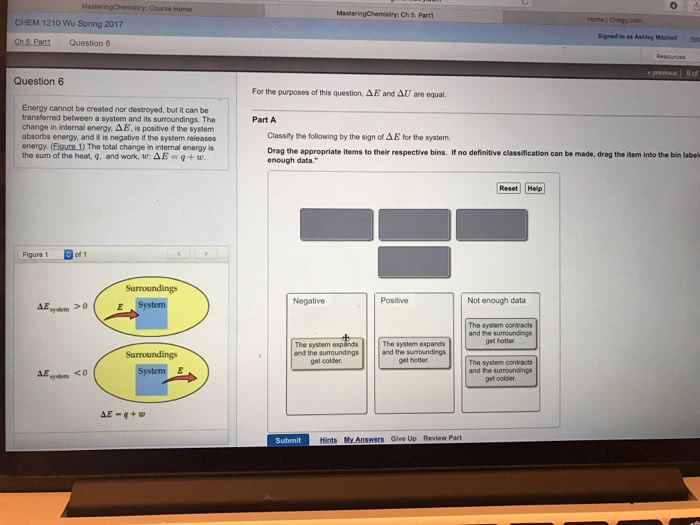
Thermodynamics: studies relationship between heat and other forms of energy. 1st law of thermodynamics: the total amount of energy in the universe is constant, although the form of energy may change. Energy cannot be created or destroyed; only transferred into a different medium. System: part of the universe in question during thermodynamic analysis. The energy change of a system ( u) is the difference between heat (q) exchanged with the surroundings and the work (w) done by the system on the surroundings. Under constant pressure p, work is de ned by the volume change of the system. Enthalpy, h = q = u + pv. In most biochemical reactions, v is incredibly small, so h = u, or heat exchange approximates work done by the biochemical system. Spontaneous processes: occur without the input of energy. If all components of a reaction are at 1m concentration. Not necessarily the concentration in question and may differ from experimental conditions in question.