BIOCHEM 3H03 Lecture Notes - Lecture 5: Spectroscopy, Crystallization, Hydrometer
Document Summary
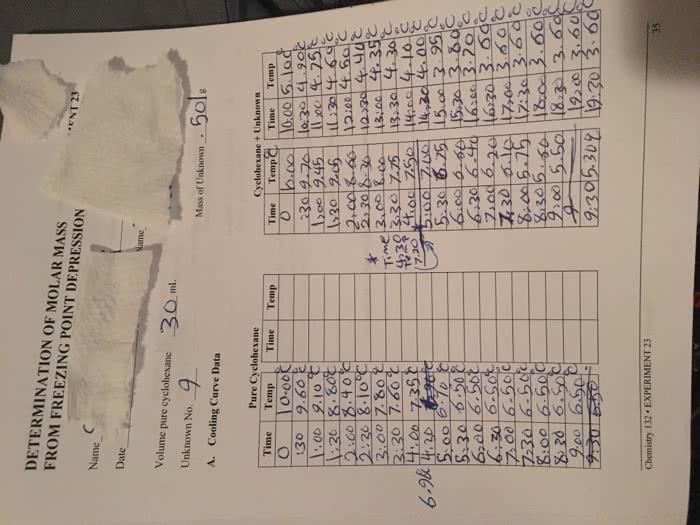
Osmometry: technique for measuring the concentration of solutes that contribute to osmotic pressure of a solution. Colligative properties: properties of a solution that depend only on the number and not the nature of the dissolved particles: examples: osmotic pressure, elevation of boiling point, freezing point depression, vapour pressure lowering. Other ways to estimate osmolality (cid:1005) (cid:1007) (cid:1006) (cid:1008) Most often done to estimate urine osmolality. Specific gravity: relates the mass of solution to the mass of an equal volume of water at 4c: density test. In most instances density and osmolality testing correlate well. Specific gravity is a quick and cheap way to estimate osmolality of urine: hydrometer, specific gravity determined using refractometry, most determined with dipstick tests chemical test which correlates with refractometry. Most abundant substances that contribute to osmolality in plasma: na and associated anions, glucose and urea. Estimated osmolality = 2[na] + [glucose] + [urea: quick way to estimate.