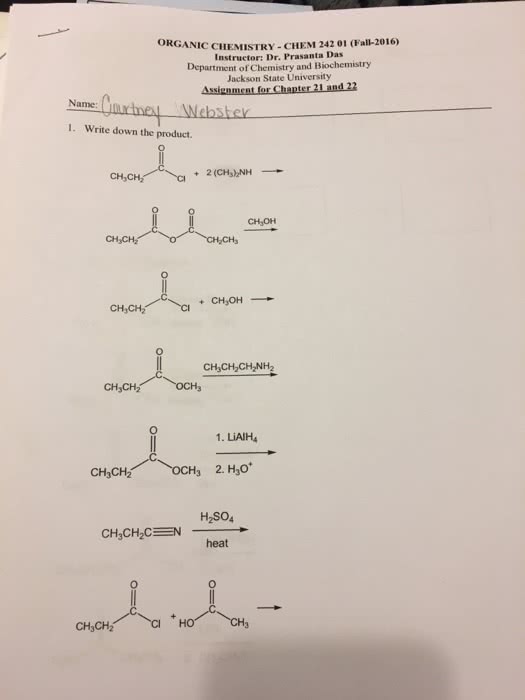
CHEM 1E03 Lecture Notes - Lecture 35: Manganese Dioxide, Hydrogen Fuel, Acid Strength
CHEM 1E03 verified notes
35/40View all
Document Summary
Solution: look for the metal that is lower in the table: anything to the right of fe: cr, zn, al, mg, sr, everything except ag, al, mg, sr. Nickel-cadmium and lithium have solid reactants and products. Consumes h+ and so42-, and its potential depends on the extent of the reaction. Reactants continuously fed into the cell while products are removed (can run continuously) Uses hydrogen gas as the reducing agent at the anode, and oxygen gas as the cathode. Acid-base equilibrium: autoionixation of water, 2h2o(l) h3o+(aq) + oh-(aq, k = [h30+][oh-] = 1. 00*10^-7. Acid dissociation equilibrium: strong acids dissociate completely, a weak acid will not completely dissociate. Solving for x" gives: 4. 2 * 10-3 use this to solve fore each entity. Trick: if the concentration of the acid is >>> ka, than you can neglect the x in the denominator: ka = x2 / 1. 00.