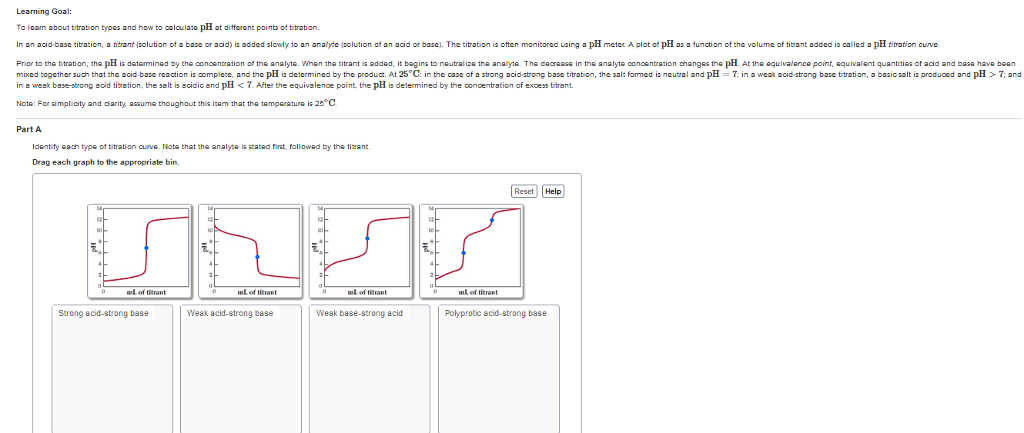
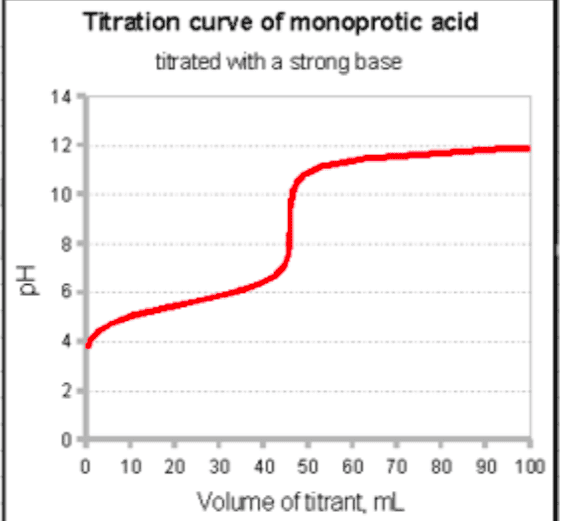
CHEM 112 Lecture Notes - Lecture 1: Acid Dissociation Constant, Equivalence Point, Burette
Document Summary
When the acid is 10 times the concentration of the conjugate base ([acid] = 10[base]), the ph is ph = pka + log[base]/10[base]= pka + log(1/10) = pka 1. The buffer region is therefore ph = pka 1. This technique, which can involve an acid -base reaction or an oxidation -reduction reaction, is known as volumetric analysis . In an acid-base titration, the titrant could be an acid and the analyte a base, or vice-versa. The end of the titration, the stoichiometric point (or equivalence point) is detected by using an indicator dye, which changes colour at the ph of the neutralization of the acid or base. Strong acids and strong bases are leveled in water to the strength of the h3o+ (aq) and oh- (aq), respectively, generated from their interactions with water. It is therefore a reaction between h3o+ and oh- which is the net reaction in a titration involving a strong acid and a strong base.