CHEM 282 Lecture Notes - Lecture 19: Leaving Group, Alcohol, Electronegativity
45 views2 pages
25 Dec 2017
School
Department
Course
Professor
Document Summary
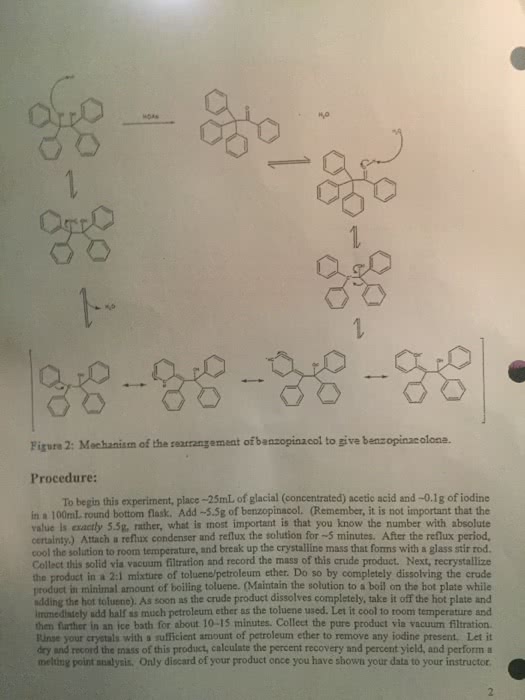
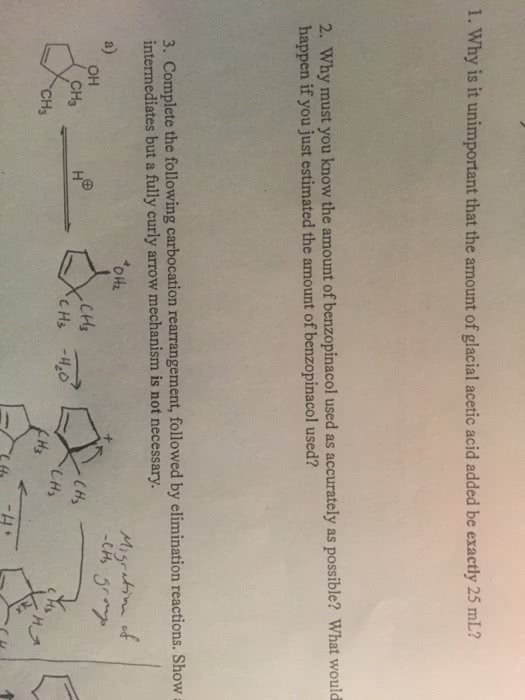
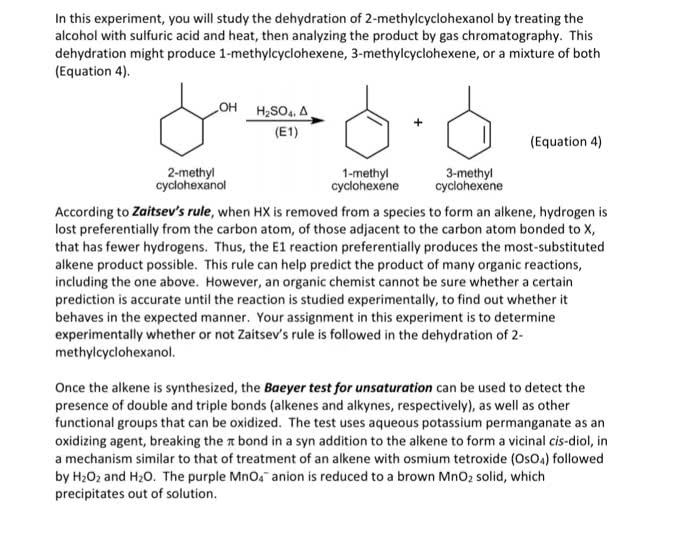
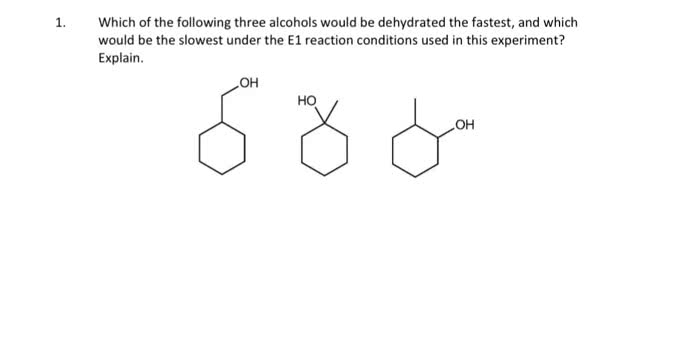
Electrons from electronegative atom attract the hydrogen; the electrons that were initially holding hydrogen become part of the double bond. The zaitsev s rule is associated with e2? elimination. With this rule, the more substituted alkene is the more stable and dominates. Oh is a good leaving group only if it is protonated. To keep it protonated, a strong and concentrated acid can be used. If it is a good base, it is probably a horrible leaving group. The dehydration mechanism is a useful e1 mechanism. Hydroxyl is protonated - this makes it a good leaving group. The substrate may be a secondary or tertiary alcohol. The formation of the carbocation is the slowest step. Carbocations can rearrange if possible to form a more stable carbocation. Good to use a solvent that solvates carbocations. The acid is regenerated in the final step.
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232