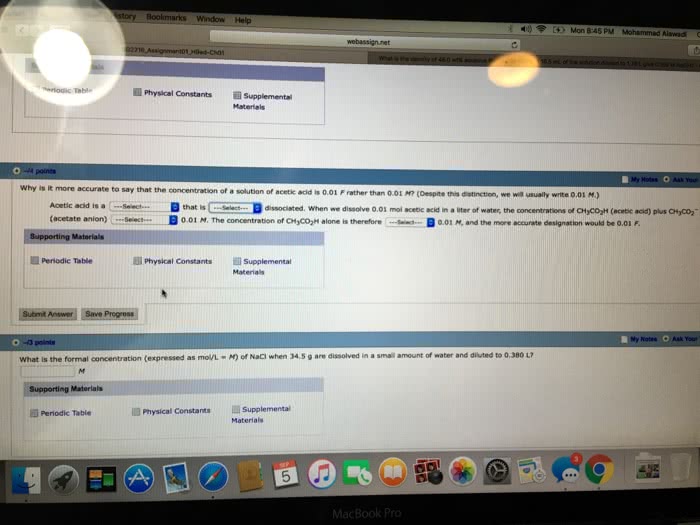
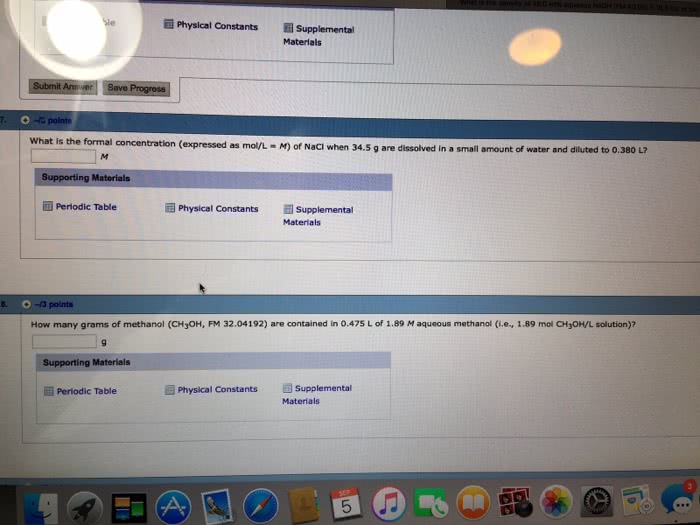
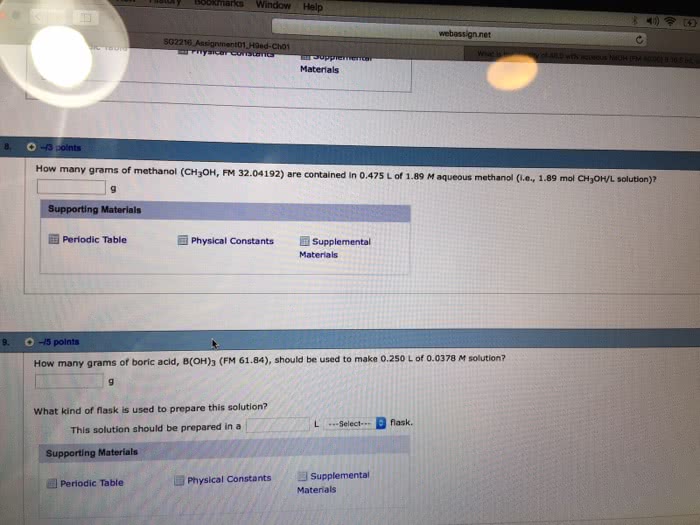
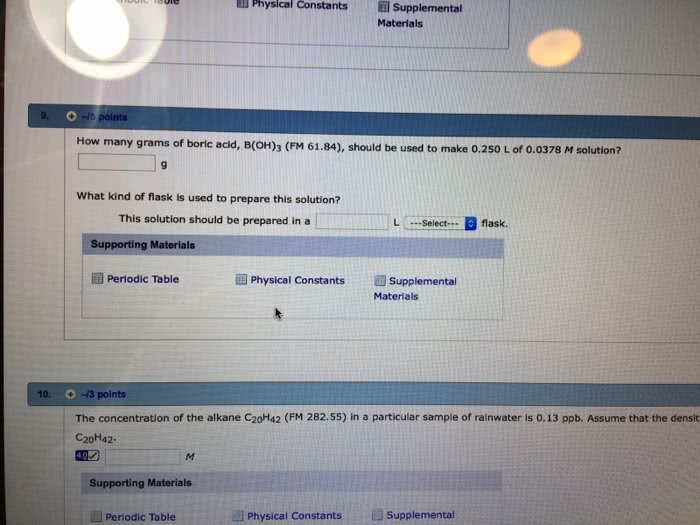
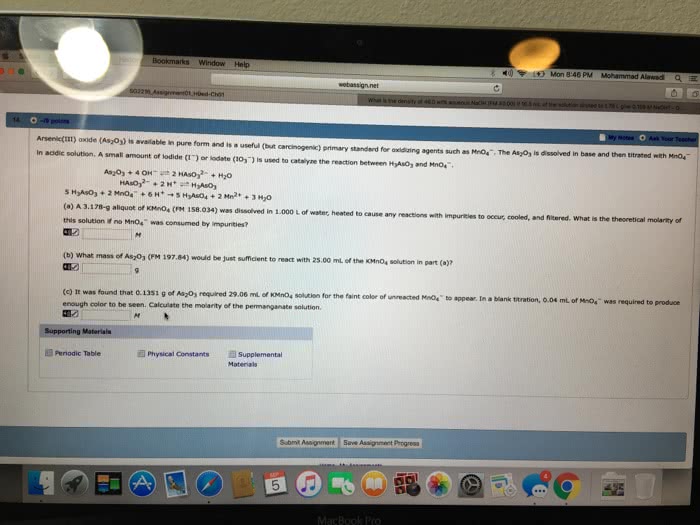
2 Part Question:
An analytical chemist is given 1.546 g of a solid coordination complex that definitely contains a Cox(C2O4)y complex (with the Co in the Co2+ oxidation state) and Li counter-ions, and possibly also water. His mission is to determine its correct empirical formula.
Part I) He takes the Cox(C2O4)y solid and chips off a 0.5103 g solid sample. He manages to dissolve this sample into a solution of about 150 mL of 2 M sulfuric acid. He then titrates a 0.101 M solution of K2Cr2O7 into this Cox(C2O4)y analyte solution. The analyte solution remains colorless until the endpoint, at which point it turns yellow. The volume of 0.101 M K2Cr2O7 solution titrated in this experiment is 11.00 mL. What is the weight % of oxalate in the original sample and what is the mmol of oxalate/100.0 g of complex?
Remember to first balance the following equation:
_ K2Cr2O7 (aq) + _ H2C2O4 (aq) + _ H+(aq) ï® _ Cr3+(aq) + _ CO2 (g) + _ H2O(l) + _ K+(aq)
a) 7.67 wt% & 87.1 mmol b) 47.9 wt% & 544 mmol c) 19.2 wt% & 218 mmol d) 57.5 wt% & 654 mmol e) 6.39 wt% & 72.6 mmol
Part II) After thinking it over, the analytical chemist decides that a precipitation would be more accurate (and easier) than spectrophotometry. Hence, he dissolves a 0.5168 g sample of Cox(C2O4)y in 100.0 mL of aqueous solution and makes sure the Co2+ is freed from the Cox(C2O4)y with the addition of acid. He then adds an excess of ammonium phosphate, (NH4)3PO4, resulting in the precipitation of a red solid powder. Once completely dried, the mass of the red solid powder of Co3(PO4)2 is measured to be 0.1356 g. Determine the amount of Co2+ in the original sample.
From the determination of the amount of Co2+ in the original Cox(C2O4)y complex and your answer for C2O42- in the previous question, which one of the following answers appears to be the full empirical formula?
a) Li6[Co2(C2O4)6]·13H2O b) Li6[Co2(C2O4)5]·18H2O c) Li3[Co(C2O4)3]·6H2O d) Li16[Co(C2O4)9]·24H2O e) Li4[Co(C2O4)3]·6H2O